Structure and Dynamics of Superconducting NaxCoO2 Hydrate and Its Unhydrated Analog
Cobalt oxide systems have been attracting increased attention recently because of their interesting magnetic and thermoelectric properties, as well as for possible analogies to colossal magnetoresistive manganite materials, or high superconducting transition temperature cuprate oxides. For the NaxCoO2 system of particular interest, the spin entropy has been found to play an essential role in the dramatically enhanced thermopower for large sodium content (x ~ 0.7), while magnetic ordering has been observed at high x, and at x=1/2 where the system is a charge-ordered antiferromagnetic insulator. At small x (~0.3), the recent discovery of superconductivity in hydrated NaxCoO2 has been of particular interest with regard to the superconducting cuprates. This is a layered system where the Co4+ ions are in the low-spin state and carry S=1/2 so that quantum effects are maximal, while the underlying lattice is triangular rather than square like the cuprates. These observations suggest that this may be the first new class of "high-TC" superconductors since the discovery of the cuprates over 19 years ago, but of course the nature and mechanism of superconducting pairing in this new class of materials is in the early stages of being addressed. The appropriate underlying model may be a Mott insulator in two-dimensions, with S=1/2 where quantum fluctuations are optimal. The Co spins would then play a critical role in forming Cooper pairs that might have triplet symmetry, or d-wave symmetry as in the cuprates. On the other hand, the traditional electron-phonon interaction may be establishing conventional s-wave pairing, with the possibility that the anharmonic motion of the hydrogen and oxygen ions might be playing a role in enhancing the superconducting properties, in a manner similar to MgB2. We have determined the crystal structure and lattice dynamics for the superconducting hydrate, and compared the behavior with the related non-superconducting compound Na0.3CoO2 [Phys. Rev. B 68, 214516 (2003)].
We have investigated the crystal structure of NaxCoO2 as a function of doping x, and related the structure to the observed physical properties. The phase diagram as a function of Na doping is shown in the figure below [Phys. Rev. B 70, 184110 (2004)]. Each Na ion nominally donates an electron to the CoO2 plane, but we see that the behavior of the Na is much more complicated than this simple picture would suggest. In particular, the Na ions occupy two different sites over a wide range of x, as shown in the structure figure, while the CoO2 is structurally robust. The system is a paramagnetic metal for x<1/2, with both the Na(1) and Na(2) sites being partially occupied, and the Na(2) being further split into a threefold site that is again randomly occupied. This is designated the H1 structure. At x=1/2 the system exhibits a special charge and orbitally ordered structure that is insulating, with the Na ions occupying ordered positions that form one-dimensional zigzag chains [J. Phys.: Cond. Matter 16, 5803 (2004)]. Recent single crystal measurements indicate that below a Neel temperature of 88 K the spins form a novel antiferromagnetic pattern within the CoO layers, consisting of alternating rows of ordered Co spins and spinless Co ions [Phys. Rev. Lett. 96, 046403 (2006)]. For x>1/2 the system returns to the H1 structure and is a Curie-Weiss metal. Around x~3/4 the Na structure transforms to the more ordered H2 structure, where the randomness in the Na(2) site is absent.
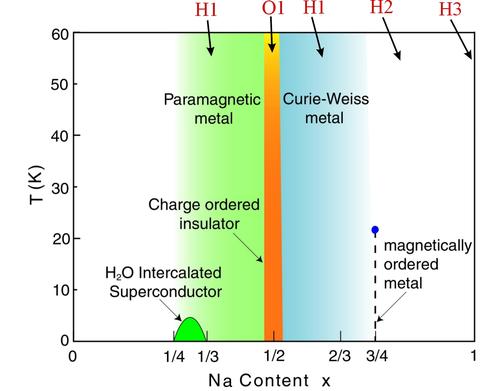
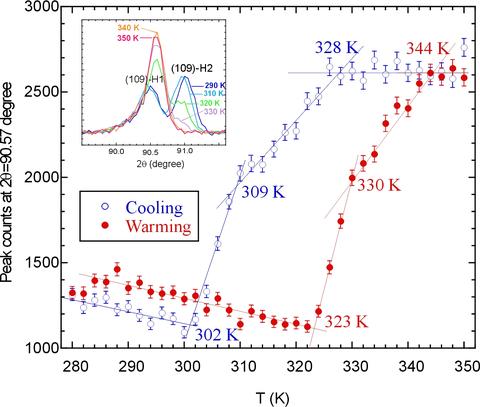
This transition can also be driven thermally as shown in the figure below [Phys. Rev. B70, 134115 (2004)], where we observe the quite unusual behavior of transforming from an ordered state at low T to a randomly occupied threefold degenerate site at elevated T. This transition is first-order in nature in both temperature and composition. Finally, with further increase of the Na composition we realize the H3 structure, where the Na(2) site is fully occupied and the Na(1) is completely vacant. The transition from the H2 to the H3 structure as a function of composition is also discontinuous in nature.
One of the amazing characteristics of this system is the discovery of superconductivity when the material is hydrated. Initially water goes into the Na layer, but then the water forms its own layer between the Na and CoO2 layers, with the c-axis expanding from 11.2 Å to 19.5 Å and the material becoming a 5 K superconductor. This is a layered system where the Co4+ ions are in the low-spin state and carry S=1/2, just like the cuprates, and the dependence of the superconducting transition on band filling is reminiscent of the cuprates. So far the highest superconducting transition is 5 K, but they are strongly anisotropic type-II materials (k~102), again, similar to the cuprates. These observations suggest that this may be a new class of "high-TC" superconductors since the discovery of the cuprates over seventeen previously. Of course, the nature and mechanism of superconducting pairing in this new class of materials is in the early stages of being addressed. The appropriate underlying model may be a Mott insulator in two-dimensions, with S=1/2 where quantum fluctuations are optimal. The Co spins would then play a critical role in forming Cooper pairs that might have triplet symmetry as in Sr2RuO4 or d-wave symmetry as in the cuprates. On the other hand, the traditional electron-phonon interaction may be establishing conventional s-wave pairing, with the possibility that the anharmonic motion of the hydrogen and oxygen ions might be playing a role in enhancing the superconducting properties, in a manner similar to MgB2.
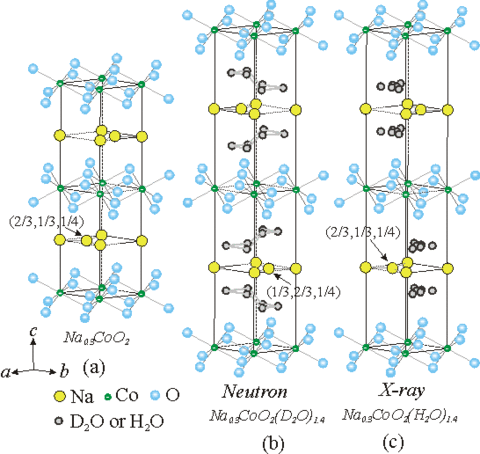
Both the structure and lattice dynamics indicate that this separate water layer has the basic structure of ice as shown on the right-hand side of the figure below. Note in particular that the structure of the Na is different than in the parent compound, with the Na shifting to the other side of the unit cell to accommodate the water. A central question is whether the water is playing an active role in the superconducting pair formation, or is simply expanding the lattice and making the system more two-dimensional in nature. So far, however, only water has been found to render the system superconducting. We have investigated both the crystal structure and lattice dynamics for three different samples of the superconducting hydrate Na0.3CoO2 ·1.4(H/D)2O, and compared the behavior with the related non-superconducting compound Na0.3CoO2. The structure of Na0.3CoO2 consists of alternate layers of CoO2 and Na and is the same as the structure at higher Na concentrations. For the superconductor, the water forms two additional layers between the Na and CoO2, increasing the c-axis lattice parameter of the hexagonal P63/mmc space group from 11.16 Å to 19.5 Å. The Na ions are found to occupy a different configuration from the parent compound, while the water forms a structure that replicates the structure of ice. Both types of sites are only partially occupied. The CoO2 layer in these structures is robust, on the other hand, and we find a strong inverse correlation between the CoO2 layer thickness and the superconducting transition temperature as shown in the figure below. The phonon density-of-states for Na0.3CoO2 exhibits distinct acoustic and optic bands, with a high-energy cutoff of ~100 meV. The lattice dynamical scattering for the superconductor is dominated by the hydrogen modes, with librational and bending modes that are quite similar to ice, supporting the structural model that the water intercalates and forms ice-like layers in the superconductor [Phys. Rev. B 68, 214516 (2003)]. Recent high resolution work using the high flux backscattering spectrometer has revealed that most of the water is static on an energy scale of 1 meV [Phys. Rev. B 75, 014505 (2007)].

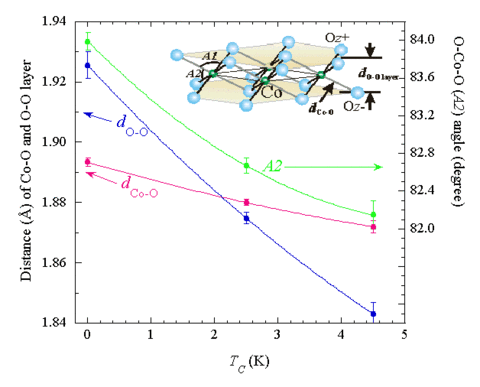
(Top) Structure for the NaxCoO2 1.4(D2/H2O) superconductor. The inset shows the D2O ice structure; for comparison the sketch in the middle shows the ice molecule superposed on the water block in the structure of the superconducting phase, demonstrating that the dimensions of the water block are close to those found in free ice. (Bottom) Interatomic distances for Co-O in the CoO2 layer, and the O-Co-O bond angle, as a function of the superconducting transition temperature TC. Note the strong dependence of TC on the bond angle A2 (defined in the inset), which determines the CoO2 layer thickness.
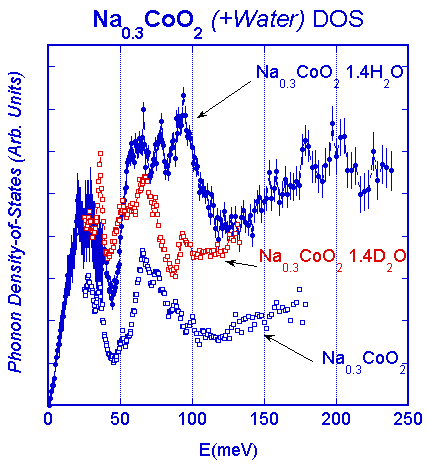
Measurements of the generalized phonon density-of-states for the parent compound, the deuterated superconductor, and the protonated superconductor. For Na0.3CoO2 the excitations divide into an acoustic band and an optic band, with a one-phonon density-of-states cutoff around 100 meV; at higher energies the scattering is multiphonon in origin. For the protonated superconducting sample there is additional strong scattering in the 50-120 meV range, which matches relatively well to the librational modes of ice, while at higher energies there are bending modes. The intensities of these bending modes have a significant temperature dependence due to Debye-Waller effects, while below ~150 meV temperature has little effect. For the deuterated sample the positions of the hydrogen modes scale with the mass. The sharp peak at 37 meV also agrees with the peak position from ice, indicating that the dynamics of the water in the superconductor is quite similar to the dynamics (and structure) of ice.
Structure and Dynamics of Superconducting NaxCoO2 Hydrate and Its Unhydrated Analog, J. W. Lynn, Q. Huang, C. M. Brown, V. L. Miller, M.L. Foo, R.E. Schaak, C. Y. Jones,.E. A. Mackey, and R. J. Cava, Phys. Rev. B 68, 214516 (2003).
Structural Transition in NaxCoO2 with x near 0.75 due to Na Rearrangement, Q. Huang, B. Khaykovich, F. C. Chou, J. H. Cho, J. W. Lynn, and Y. S. Lee, Phys. Rev. B 70, 134115 (2004).
Low Temperature Phase Transitions and Crystal Structures of Na1/2CoO2, Q. Huang, M. L. Foo, J. W. Lynn, H. W. Zandbergen, G. Lawes, Y. Wang. B. Toby, A. P. Ramirez, N. P. Ong, and R. J. Cava, J. Phys.: Cond. Matter 16, 5803 (2004).
Coupling Between Structural and Electronic Degrees of Freedom in the Triangular Lattice Conductor NaxCoO2, Q. Huang, M. L. Foo, J. W. Lynn, B. H. Toby, R. A. Pascal, H. W. Zandbergen, and R. J. Cava, Phys. Rev. B 70, 184110 (2004).
Neutron Scattering Study of Novel Magnetic Order in Na0.5CoO2, G. Gasparovic, R. Ott, J. H. Cho, F. C. Chou, Y. Chu, J. W. Lynn, and Y. S. Lee, Phys. Rev. Lett. 96, 046403 (2006).
Diffusion of Water in the Na0.3CoO2·1.4H2O Superconductor, Victoria Garcia Sakai, Eugene Mamontov, J. W. Lynn, L. Viciu, and R. J. Cava, Phys. Rev. B 75, 014505 (2007).
 Recent Publications Return to Home page
Recent Publications Return to Home page

