Inkjet Printing of Medicines
Summary:
Drop-on-Demand (DOD) inkjet printing allows precise deposition of picoliter (pL)-sized droplets of solutions containing inorganic and organic materials to support a variety of critical programs at NIST. In one of the programs, we are currently exploring the possibility of using the inkjet printer as a modality to prepare personalized medicines with the emphasis on preparing the “right drug at the right dose at the right time” based on the patient’s needs combined with the convenience of real time preparation. This of course requires tools for characterization and validation, and the inkjet printer currently assists in the preparation of quantitative test materials for advanced surface analysis tools such as imaging mass spectrometers.
Description/Details:
Drop-on-Demand (DOD) inkjet printing allows precise deposition of picoliter (pL)-sized droplets of solutions containing inorganic and organic materials. We have developed an expertise in quantifying and controlling the deposition process and have applied this knowledge to the direct printing of pharmaceutical ingredients onto multiple delivery mechanisms (for example, Figure A shows the printed patterns of NaCl on an edible paper). Using a range of optical and mass spectroscopic techniques, we have confirmed the spatially localized deposition of numerous drugs and medicines at quantities up to milligram (mg)-levels.

Quantitative Test Materials for Advanced Surface Analysis Tools
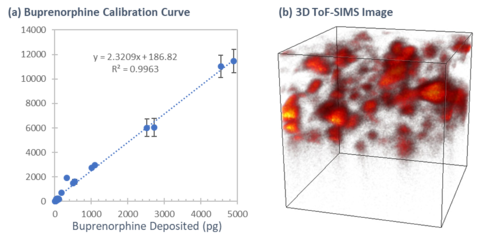
By having control of the deposition process, key parameters such as the spatial distribution, particle size, and amount of the analyte could be adjusted to produce test materials to validate the effectiveness of various characterization tools at NIST. In one example shown in Figure B, an opioid called buprenorphine was printed at discrete amounts to create a calibration curve for an instrument called the time-of-flight secondary ion mass spectrometer (ToF-SIMS). It showed that the instrument has the dynamic range to accurately quantify this material in the femtogram to nanogram range inside a small volume, concentrations that are relevant for drug delivery systems. In addition, we were able to translate this test material data to obtain quantitative information from an actual commercial product containing buprenorphine.
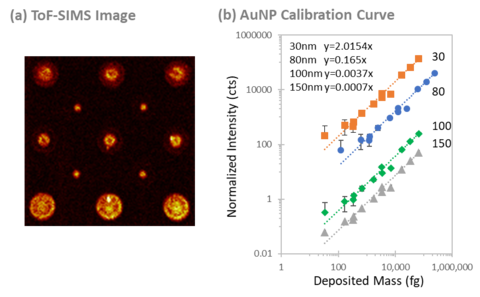
In another example, similar methodology was used to print discrete amounts of differently sized gold nanoparticles onto gelatin (Figure C), which was chosen as a matrix-matched surrogate for a biological system. A linear relationship could be obtained using ToF-SIMS, which showed that even inorganic particles could be quantified using this technique, at toxicologically relevant concentrations of a few (femtograms) to thousands of nanoparticles (picograms) per deposit. This showed that pharmaceutical ingredients tethered to the nanoparticles could also be quantified using this methodology. In the near future, we plan to quantify gold nanoparticles embedded in organisms to elucidate the toxicological pathway of these particles.
Select Publications:
Muramoto, S, Gillen, G, Collett, C, Zeissler, CJ, Garboczi, EJ. ToF‐SIMS depth profiling of oral drug delivery films for 3D visualization of active pharmaceutical particles. Surf Interface Anal. 2020; 52: 76– 83. https://doi.org/10.1002/sia.6707
Muramoto, S, Bennett, J. Inkjet Printing of Gold Nanoparticles onto a Biologically Relevant Matrix to Create Quantitative Test Materials for Time-of-Flight Secondary Ion Mass Spectrometry. Surf Interface Anal. Accepted 6/16/2020
Contacts
-
(301) 975-5997

