Taking Measure
Just a Standard Blog

From left to right: Brenton Knuffman, Adam Steele and me with our MOTIS ion source.
As a research scientist, I often face a difficult problem when discussing my work with family or friends. Everyone seems initially fascinated that I am a physicist, but if I start to talk about what I actually do, the going quickly gets tough. How do I make cold atoms and ion beams understandable and seem—at least a little bit—relevant and important? One thing that helps is to speak about it with great enthusiasm. That works for a little while because enthusiasm is contagious. But ultimately, people want to know how their lives might be affected or whether there is an economic benefit from the work.
Most of the time the economic benefit of fundamental research comes slowly, and it’s difficult to pinpoint how any one piece of work contributes. Sure, just about everyone knows that science is important for the nation and can accept that the technological developments they have come to enjoy are a direct outgrowth of the worldwide scientific research endeavor. But it’s pretty hard to look at the 2,585 papers published in Physical Review Letters in 2017 and say, “Oh yeah, I see how it works.”
On the whole, we scientists are OK with that because we love what we do and derive great satisfaction from solving complicated, difficult problems and shedding light on baffling new phenomena. But every now and then, things come together in a way that really brings us closer to an immediate practical application. Sometimes, our work leads us to an invention.
Inventions are funny things. It’s hard to tell people, “go invent something,” and as researchers in a government lab, it’s not our main job function to create inventions. But the research process is an inherently creative one, and inventiveness is extremely useful for solving the important problems we are working on. Perhaps it’s not so surprising that inventions pop up pretty often at a lab like the National Institute of Standards and Technology (NIST). When they do, it’s important for us to recognize them, protect them for future licensees, and then transfer the technology as smoothly as possible into the commercial sphere.
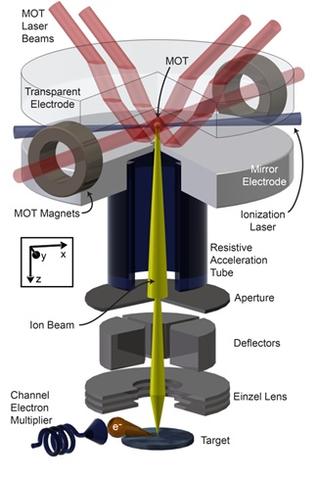
My involvement with bringing an invention from concept to patenting and ultimately licensing has been one of the most rewarding experiences of my career. And I don’t mean financially rewarding (although there are some small royalties involved). No, what’s really rewarding is seeing an idea grow from a small seed into a first demonstration and then seeing the technology launch into a bona fide business.
The research that led to an invention, in this case, was the exploration of laser cooling of atoms as it relates to nanotechnology, an area of study that encompasses just about every engineered thing 100 billionths of a meter or less in size—from transistors on a computer chip to particles used for the treatment of tumors. But laser cooling, you might ask? Yes, aiming a laser at a cloud of atoms doesn’t necessarily cook them as one might think; it actually can make them much colder than interstellar space. Since the early 1990s, I have been looking at ways these really cold temperatures could be harnessed to work with materials on the smallest of length scales. My approach has been based on a kind of precision argument: If you can precisely control a property of a material such as its temperature (which is in a general sense a measure of how randomly something is moving, where colder = less random), then there are often ways to translate this into control of another property such the material’s position (it’s easier to put something where you want it when it’s not moving so randomly).
In the mid-2000s, after developing ways to make nanostructures with laser-focused atomic deposition, metastable atom lithography, and deterministic single-atom sources, my colleagues and I were pondering what would be the best way to get cold atoms into a specific location on a surface. Making them electrically charged, or ionizing them, seemed like a good choice because then we could then use electrostatic lenses—like those used in an electron microscope—to accelerate and focus them. We did some back-of-the-envelope calculations and realized pretty quickly that an ionized cloud of laser-cooled atoms would make for an extremely fine, nanometer-scale focused beam.
As it turns out, such sharply focused ion beams are extraordinarily useful and have a firm place in the suite of tools assembled by any nanotechnology lab. They can act like sandblasters and carve tiny patterns into materials, or they can cause local chemical reactions that we can use to build up nanoscale features. This makes them a tool of choice for the semiconductor industry, where they are used to perform “circuit-edit,” or in situ rewiring of chips to test circuit modifications. When coupled with a mass spectrometer, they can knock material off a surface and measure its chemical constituents with amazing sensitivity. They are also essential for transmission electron microscopy because they allow researchers to cut out nanometer-thin slices of material for study. Even used like an electron microscope, which “sees” by bouncing electrons off nanoscale objects, they show certain advantages. Altogether, the commercial sale of focused ion beam systems constitutes a $300 million to $600 million annual market.
The commercial technology for producing a focused ion beam had been stable for several decades. At the time, nearly all focused ion beams were using liquid gallium as their ion source, which produces a few-nanometer-wide spot of gallium ions when accelerated to an energy of about 30,000 electron volts (30 keV). While this source had many advantages, its limitations were beginning to be felt as researchers delved deeper into the nanoworld. Sometimes a 30 keV gallium ion beam causes too much collateral damage as it strikes a surface, so a lower beam energy or a different element might be more desirable for some applications. Most critically, for the semiconductor industry at least, demands for spot sizes smaller than what gallium can produce were emerging with the industry’s inexorable march toward making smaller and smaller devices.
Realizing the potential of our idea to address some of these issues, we drafted a patent disclosure and began working on demonstrating the principle of our new ion source, which we dubbed MOTIS, for Magneto-Optical Trap Ion Source. We also began conversations with potential licensees and soon entered into a cooperative research and development agreement with a focused ion beam manufacturer. For our first approach, we used lithium ions. Laser cooling lithium atoms is relatively straightforward and well-documented in the literature, and we knew their low mass would be well-suited to some of the microscopy applications we were contemplating.
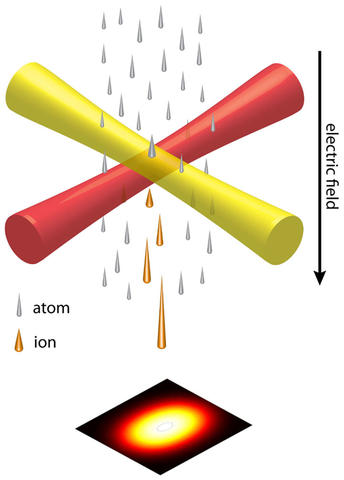
However, not all inventions follow a smooth, straight path to commercial adoption. We were quite successful in constructing the lithium ion source, realizing spot sizes as small as 27 nanometers and demonstrating a number of new forms of microscopy. But our industrial partner found the results did not quite meet their expectations and graciously bowed out of the project.
As that collaboration was winding down, the two postdocs on the project, Adam Steele and Brenton Knuffman, and I developed a next-generation version of the source that had dramatically better performance. Called LoTIS, for Low-Temperature Ion Source, the new approach used laser cooling to create a slow, intense beam of cesium atoms, which we ionized after compressing and cooling them further with carefully designed magnetic fields and more laser beams. We filed a patent for this source as well.
Adam and Brenton, being of entrepreneurial spirit, decided to launch their own startup company to develop the LoTIS technology further once they finished their postdoc terms. They took courses in small business development at Johns Hopkins University and entered and won the University of Maryland Business School business plan competition. After using a Small Business Innovation Research (SBIR) phase I grant they won from the National Science Foundation to demonstrate the value and feasibility of the invention, they proceeded to win an SBIR phase II grant.
The result has been the demonstration of a new cesium ion source with performance 24 times better than a gallium liquid metal ion source. Adam and Brenton’s company now has one additional employee and is up and running in its own space. They’re continuing to develop the source to bring it to the point of full commercialization and are receiving funding from a number of industrial partners.
As a federal employee, I’m not allowed to own any part of, or participate actively in, the operations of their company due to ethics rules. While to some people this might seem like it would be a disappointment, it’s not really a problem for me. I am just extremely happy to see an idea hatched in my lab develop into a full-fledged small business. Because the ongoing development of the ion source matches part of NIST’s mission to develop new measurement tools, we continue to work together under a cooperative research agreement.
So, now I have a story to tell my family and friends when they ask me about my work. I still get glazed eyeballs if I’m not careful and go too deeply into the details, but I feel like I’m making a small difference in helping build the nation’s technology infrastructure, and that’s something I can tell people with pride.






Great work and great outreach and education efforts.