Taking Measure
Just a Standard Blog
Experts Next Door: How a Public-Private Partnership Benefits the Regenerative Medicine Community
I’m a scientific program manager at the Standards Coordinating Body (SCB), a private nonprofit organization devoted to advancing standards for regenerative medicine, an emerging field that includes cell therapies, immunotherapies, tissue engineering and other next-generation treatments. Presently, or rather, until the coronavirus intervened, we share office space on the National Institute of Standards and Technology (NIST) campus in Gaithersburg, Maryland.
A few months ago, I was searching for experts who worked with organ-on-a-chip systems, which use cells flowing through tiny channels to simulate organs and organ systems, for the SCB-coordinated Microphysiological Systems project, which, among other things, is seeking to create a common vocabulary for describing these devices’ capabilities. I was chatting with the NIST researcher next door. When she asked what I was working on, I told her. She laughed and said, “Well, that’s what I do!” We walked down to her laboratory, and she showed me her work.
Not only did she join my working group as a subject matter expert, but she also pointed me toward several other experts I likely would not have identified without her help. Being able to see where she worked and ask her questions helped me conceptualize potential areas for standardization and provided a level of understanding I couldn’t get simply by reading journal articles. All that because I needed to stretch my legs! Similar opportunities for collaboration happen all the time in our shared workspace (though now virtual), helping us to work more effectively and ensure that the standards we coordinate are as valuable as possible to the regenerative medicine community.
Pairing Complementary Skills to Accelerate the Pace of Standardization
SCB and NIST formed a partnership in 2016 to jointly advance standards for the regenerative medicine community. A driving force behind the partnership was the 21st Century Cures Act, which directs the U.S. Food and Drug Administration (FDA) to coordinate and prioritize the development of standards for advanced therapies in consultation with NIST and industry stakeholders.
Because at their heart most standardization needs are measurement needs, NIST — a federal laboratory dedicated to the science of measurement and a nonregulatory agency of the U.S. Department of Commerce — is strongly positioned to support the development of standards, thanks to its specialization in measurement science and data. But NIST also recognizes that standard creation can proceed more efficiently with cooperation between private and public entities.
By collaborating with SCB, NIST has been able to benefit from SCB’s close relationships with the companies and small manufacturers most involved in the rapid advancement in the field of regenerative medicine. SCB can also help coordinate between NIST and international entities, ensuring NIST is aware of what is happening outside of the U.S. and knows what international regenerative medicine standards projects would be useful to U.S. interests.
Similarly, SCB benefits from NIST’s reputation in the scientific community. NIST’s credibility enabled us to more readily form new partnerships when SCB was first becoming established as an organization and continues to support our growth as we pursue our mission of accelerating standards development for the regenerative medicine community.
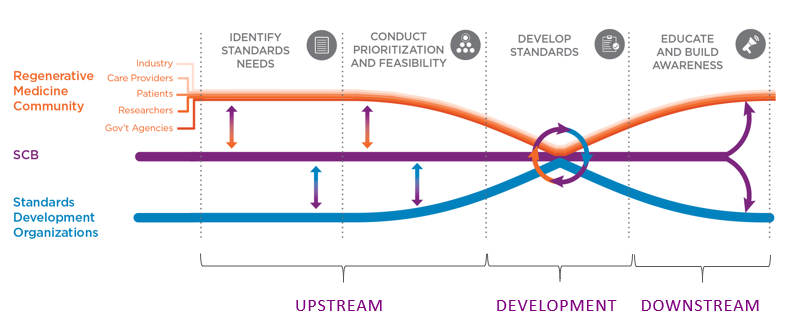
Using Methodological Thinking to Anticipate Standards Needs
NIST is probably better at methodological problem solving than any other organization I’ve ever been involved with; by analyzing scientific issues in regenerative medicine in a structured manner, its researchers are often able to anticipate what the community will need before members even realize it. At SCB, we have incorporated NIST’s methodological thinking into our own procedures in the development of new standards to help us more effectively identify standards needs.
Because a common challenge for the regenerative medicine community is the duplication of effort that can occur when multiple organizations try to tackle the same technical problem independently, these early insights into standards needs help us bring groups together preemptively to solve problems before efforts begin to diverge. For example, something as small as how labels are printed for samples, without a common effort, can cause a clinic to need multiple printers and labels that would all put information in different places and require the technician to receive multiple trainings to decipher each different type of label. At the clinic level this is costly, time-consuming and can lead to errors. Once scientific data becomes locked up in proprietary formats, it can be much more difficult to share, so getting groups to work together to standardize things like sample labels pre-competitively speeds the process of standards development by a significant degree. The standards that come out of these collaborations benefit the whole community, helping accelerate the development of new therapies by making foundational scientific knowledge more accessible to organizations of all sizes.
Assessing Real-World Use Cases to Develop More Valuable Standards
We work closely with NIST’s Biosystems and Biomaterials Division, run by Sheng Lin-Gibson. The division’s work spans from the quantification of DNA to cell and gene therapies to tissue-engineered products. These in-house experts use — and in many cases even create — cutting-edge regenerative medicine methodologies, giving them unique insight into real-world applications. Lin-Gibson knows the field better than almost anyone and can offer critical input on how standards may be used by individual organizations.
To develop a standard from start to finish, regenerative medicine experts volunteer hours of their time to research, draft and discuss the standard — time that ultimately goes to waste if a standard isn’t used by the broader community. Making sure we use their time wisely is a paramount concern for SCB.
Lin-Gibson helps us to work backward when thinking about standards, asking ourselves who will use the standard and how it will help them. This approach helps us to not only make better quality standards, but also to avoid focusing the limited time, attention and resources of the community on advancing standards that are unlikely to be used.
Working Together to Set Standards Up for Success
Many of the problems in standardization are, at their heart, measurement needs, which is part of what makes NIST an ideal cooperative partner for our small nonprofit. Having access to the experts at NIST — to contribute measurement expertise, help us anticipate the standards needs of the regenerative medicine community, and offer insight and feedback on real-world standards applications — strengthens and accelerates our work.
We have already seen the benefits of this partnership in action. Projects coordinated by SCB can take less than one year to undergo the pre-development steps of standardization — identification of standards needs, prioritization and assessment of feasibility. Projects without this support can take up to six years to complete these steps. The pre-development stage is critical for identifying the standards that will be most valuable and successful to the community before advancing them to standards developing organizations (SDOs), the last step before they become available to the regenerative medicine community. And the sooner the community can begin availing themselves of these standards, the faster their products can start making a difference for the patients who need these potentially life-saving regenerative therapies.
We could not have been this successful in accelerating this early assessment phase without our strong working relationship with NIST to help us better understand the needs of those in the field. I deeply appreciate the opportunity to work with these next-door experts to exchange ideas, solve problems and answer critical questions to ensure we can create the best possible standards for the regenerative medicine community.
Get Involved
If you’d like to learn more about our work and how you can participate in regenerative medicine standards development, please czander [at] regenmedscb.org (drop me a line).
About the author
Related Posts
Comments
I am interested in your research.
Collected stem cells as an RN in Apheresis Unit for over 25 years.






I am interested to join on reseaŕch