Summary
Microscopy images of cells are often used in cell biology to qualitatively identify features of interest and to discern differences between treatments. Images are also sometimes used to quantify gross morphological features. However, images are less often used to obtain quantitative intensity data of intracellular signaling species due to the paucity of validated image-based quantitation protocols. As a demonstration of the general approach to develop and validate quantitative, cell-based immunofluorescence assays, we successfully developed a methodology to quantify the relative concentration of diphosphorylated myosin light chain within single cells. This methodology is being extended to develop staining procedures for other key intracellular signaling targets.
Description
Analysis of signaling events within individual cells with microscopy imaging has several advantages over non-imaging techniques. The principal advantage of imaging cytometry over plate reader assays and even other single cell techniques such as flow cytometry is the potential for collection of morphological and spatial information over subcellular and supracellular length scales. This can be an advantage for cell based assays for drug cytotoxicity screening where even relatively simple morphological measurements, such the area of the nucleus, have been shown to be predictive of clinical outcome. Imaging cytometry can collect spatial information on several hundreds of adherent cells, as well as provide signal quantification in single adherent cells and quantification of differences between cells in the population. We examined the feasibility of developing a quantitative, cell-based immunofluorescence assay for the phosphorylation of myosin light chain, myosin being an important intermediary of many vital cellular processes, including cell division.
To take advantage of the benefits of microscopy, it was important to preserve the overall morphology of the specimen while also obtaining an optimal signal for myosin light chain phosphorylation. We first used overall cell morphology as the qualitative criteria to screened several fixation techniques. We tested four different fixation reagents: ethanol (EtOH), methanol (MeOH), acetone (Ac), and paraformaldehyde (PFA). PFA best preserved cell morphology while the organic solvents ethanol, methanol, and acetone, gave variable results as fixatives and in some cases caused cells to round up upon addition of any one of the three solvents (data not presented). PFA was selected as the fixation reagent, and four different permeabilization methods were screened to select the optimal permeabilization strategy. After fixation by PFA at room temperature, cells were permeabilized with methanol (PFA+MeOH, 4°C), ethanol (PFA+EtOH, 4°C), acetone (PFA+Ac, 4°C) or triton X-100 in PBS (PFA+TX100, room temperature). We also tested a simultaneous fixation and permeabilization procedure by including 0.5% triton X-100 in the PFA solution (PFATX, room temperature). The presence of phospho-MLC antibody staining along well-defined f-actin stress fibers was used as the criterion for evaluating the permeabilization method. Each of the different permeabilization techniques resulted in distinct differences in the staining patterns. Based on the above criteria, PFATX was found to be optimal among the methods tested to preserve myosin light chain phosphorylation and actin stress fibers.
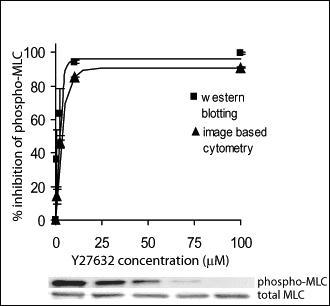
Using this specimen fixation technique, myosin light chain phosphorylation was measured as a function of increasing concentrations of a drug that inhibits MLC phosphorylation and resulting values were compared with similar measurements done by Western blotting and found to be equivalent. The assay was further validated by an additional method stood perturbed MLC phosphorylation, including changing ECM density, activating myosin phosphorylation, and performing the assay in a different cell type. A key component of this activity was the availability of a previously developed infrastructure at NIST to label and segment cell bodies using a high contrast stain, and instrumentation for automated microscopy.
Major Accomplishments
- Developed a sample fixation and staining technique that results in optimal staining for MLC phosphorylation within cells, in an antibody-based assay
- Validated the assay against Western blotting in multiple cell types, and under multiple conditions that perturbed MLC phosphorylation.
Additional Technical Details
Bhadriraju, K., Elliott, J.T., Nguyen, M., Plant, A.L., 2007, Quantifying myosin light chain phosphorylation in single adherent cells with automated fluorescence microscopy, BMC Cell Biology, 8:43.

