Summary
Ninety percent of clinical trials fail because new drugs are often not effective or turn out to be more toxic than estimated with data from animal trials. We are developing microphysiological systems that are mimics of parts of the human body and have the potential to better predict which drug candidates will be successful in first-in-human trials. Microphysiological systems (MPS) are microfluidic cell-culture devices that house mimics of human organs made from primary or stem cell sources as well as a circulatory system that recirculates drugs and drug metabolites. Testing drugs with MPS instead of animals could save billions of dollars and time, because data from MPS could identify compounds that won’t be successful. However, despite years of development, current MPS are not ready to realize their full potential role in drug development. The devices can be difficult to assemble and use, and they also do not yet capture the full complexity of the human body. Our goal is to build an MPS that mimics the body more fully and that is easy to operate.
Description
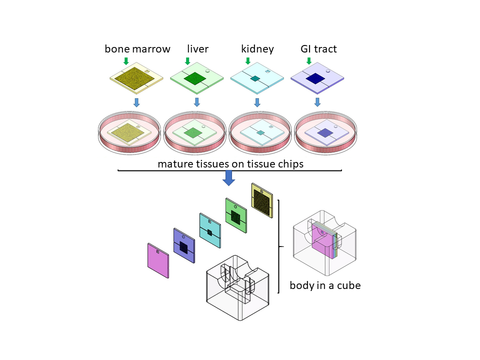
Fig: 1. A pumpless body cube with four tissues (bone marrow, liver, kidney, and GI tract) enables drug testing and toxic metabolite detection under conditions with near-physiological amounts of blood surrogate.
We are developing tissue chips and multi-tissue microphysiological devices that support the culture of multiple tissues with physiologically relevant connections and streams of cell-culture medium. The goal in this project is to construct a device that realistically mimics human metabolism as a means to reliably predict the efficacy and toxicity of drugs. Our focus is on a design that will reduce the amount of liquid (blood surrogate) needed to operate the device to near-physiological levels. We are achieving that goal by developing pumpless systems, reducing the volumes of interconnecting microfluidic channels, as well as by reducing the volumes of any valve components. Using those strategies, we have developed a pumpless system for the culture of healthy and activated endothelial cell layers, and a body cube that is capable of the co-culture of four tissues (Fig. 1). We are currently testing the devices to determine the effects of low liquid levels on the long-term culture of tissues. We are also developing mathematical models of drug metabolism inside the devices and ways to translate the data obtained with the devices to predictions for humans. The impact of MPS devices with near-physiological liquid levels is that they will predict drug action in patients with much higher reliability because drug metabolites in the system will be produced at near-physiological concentrations.

