Description

Now Available
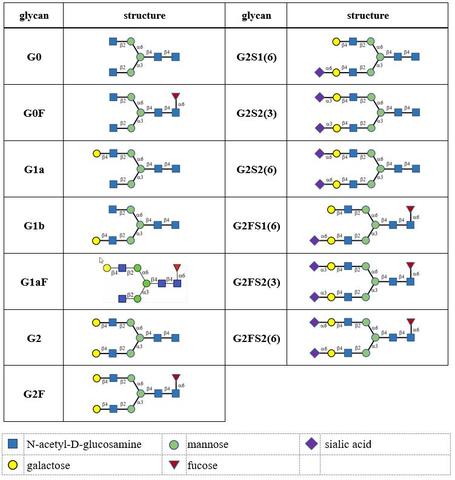
A glycan-based NIST Standard Reference Material (SRM 3655 Glycans in Solution) supports the biomanufacturing and protein therapeutics communities with a set of 13 individually-bottled, pure glycoforms including those most commonly observed as N-linked glycans on therapeutic monoclonal antibodies.
SRM 3655 is specifically beneficial for biotherapeutics development and testing of biosimilars, encouraging traceable quantification of monoclonal antibody glycosylation in regulatory submissions. Potential uses of this SRM include calibration of measurement systems, daily QC assurance testing, stability testing, interlaboratory comparisons, and for benchmarking new measurement technologies. These compounds were synthesized through established chemoenzymatic processes, purified, and certified for concentration. This quantitative glycan material enables accurate, SI-traceable quantification of antibody glycosylation, and can be used to assess quantity, identity, or stability between labs, between production lots, or over time.
Our focus is on developing standards and measurement tools for supporting the needs of the biopharmaceutical community. We aim to encourage quantitative accuracy throughout the process validation and design stages for biomanufacturing of monoclonal antibody (mAb) drug products, and ensure the availability of traceable, consistent standards for unbiased assessment of biosimilars. During mAb development, a number of significant modifications to a drug product can occur, potentially affecting the efficacy, stability, and/or safety of the drug formulation. These critical quality attributes (CQA) can be defined, measured, and monitored to ensure that a drug product remains within acceptable quality limits. One of the most important CQAs associated with biotherapeutics development is the glycan profile of the mAb. Characterization of the identity and the quantity of glycans found on a recombinant mAb are well-documented to affect biological activity and should be monitored to ensure product consistency.
Currently, glycan profiles are characterized within the biomanufacturing community by comparing new drug lots to earlier release lots of the same product. This is done through a semi-quantitative approach that may be subject to measurement biases, and which can affect quantitative accuracy. An independent, stable, and traceable Standard Reference Material (SRM) is not currently available to underpin the measurement validation steps of recombinant mAb production. However, we are currently developing just such an SRM to meet the measurement needs related to these very important therapeutic products. Highly purified unmodified (no derivatization) glycan standards, representing the most abundant and commonly identified N-linked glycans found on recombinant mammalian immunoglobulin-G mAbs, will be used to develop the NIST SRM 3655.
As the field is moving toward quantitative measurements, standards will become essential for ensuring traceable, unbiased quantification. NIST is focused on supporting the measurement needs of the glycosciences community and welcomes input on development of additional glycan-based SRMs.

