Summary
Chipscale electronic devices such as field-effect transistors (FETs) are being developed for biochemical applications ranging from clinical diagnostics to biomanufacturing and food security. A key advantage that distinguishes this approach from others is the ability to directly transduce charge, which eliminates the need for specialized labeling and markers. Despite their flexibility and scalability, the commercial adoption of these technologies has been extremely limited. We are developing an electronic measurement platform with a focus on improving sensitivity, resolution and stability to foster their broad adoption in fundamental biophysics at multiple spatial scales (e.g., to understand protein-ligand interactions in small molecular ensembles, quantify enzyme activity regulation, measure cellular function, etc.) and for clinical applications (e.g., diagnostic testing, drug discovery, etc.). To achieve this goal, we develop state-of-the-art electronic devices, realize measurement configurations to maximize performance and flexibility, design application-specific biotic interfaces, and utilize theory for robust interpretation.
Description
We develop measurements that leverage electronic signal transduction using FETs to maximize sensitivity and improve the resolution of biomolecular measurements. The techniques allow direct charge transduction during molecular interactions to quantify fundamental biophysical processes. Critically the methods support measurements of molecules at extremely low concentrations and in very small sample volumes. A key feature of our approach is that signal transduction is performed remotely from the sensing surface allowing reuse of the detection electronics and to allow the development of a library of sensors to support multiple applications as part of a modular measurement platform (see figure).
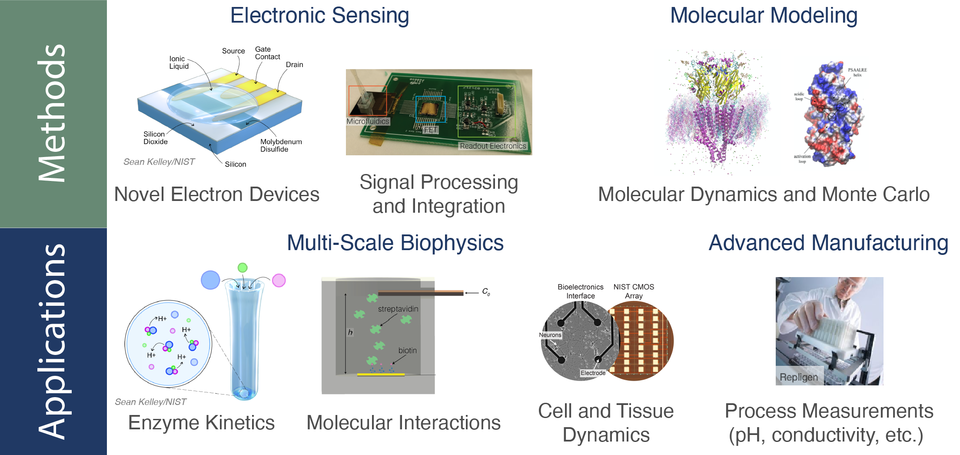
We design, fabricate and optimize high-performance signal transduction that leverages layered 2D materials such as MoS2 or graphene to realize FETs with extremely high voltage sensitivity and with high resolution. Our approach has allowed the detection of solution pH with a resolution of 300 parts per million, or ≈ 100-fold better than commercial devices. In collaboration with the NIH, we applied our techniques to the quantification of the enzyme Cdk5 that is implicated in Alzheimer’s disease. The hydrolysis of adenosine triphosphate (ATP) is critical to the function of Cdk5 and results in a small change of the solution pH that acts as a reporter of its function. A critical challenge is performing measurements at or near physiological values to evaluate enzyme function under naturally occurring conditions. Our high-performance FET-based transduction allows such measurements by enabling the quantification of small changes in the enzyme activity, and the effect of drug molecules in restoring normal enzyme function and potentially reversing the effects of disease.
We also apply the signal processing and circuit integration techniques we developed for custom built 2D material-based electron devices to conventional Si-based complementary metal oxide semiconductor (CMOS) devices. This allows us to improve the sensing resolution of both commercially sourced devices (e.g., n-channel FETs) and purpose-built CMOS devices to allow broad adoption of high-performance electronic measurements for biosensing. This approach is critical to ensure rapid scalability of biosensing using mature silicon-based technologies.
We heavily rely on this latter approach when measuring fundamental biophysical processes such as the dynamics of protein-ligand interactions on surfaces, porins in membranes, measuring nucleic acid constructs, and quantifying the function of cells in culture. The ultimate impact of our approach is the ability to rapidly adopt our platform to measure diverse biophysical systems across spatial and temporal scales.
Research Opportunities
Please contact Dr. Arvind Balijepalli at arvind.balijepalli [at] nist.gov (arvind[dot]balijepalli[at]nist[dot]gov) to inquire about postdoctoral and collaborative opportunities.
Postdoctoral opportunities are available through a National Research Council (NRC) Fellowship, a 2-year program with application deadlines of February 1 and August 1 each year.
Additional information about the specific opportunities and the program are below:
- Dr. Balijepalli research opportunity description.
- NIST NRC Research Associateship Program Information.
Selected Publications
- Le, S. T. et al. High-performance dual-gate graphene pH sensors. Appl. Phys. Lett. 120, 263701 (2022).
- Le, S. T. et al. Rapid, quantitative therapeutic screening for Alzheimer’s enzymes enabled by optimal signal transduction with transistors. Analyst 145, 2925–2936 (2020).
- Guros, N. B., Balijepalli, A. & Klauda, J. B. Microsecond-timescale simulations suggest 5-HT–mediated preactivation of the 5-HT 3Aserotonin receptor. P Natl Acad Sci USA 117, 405–414 (2020).
- Le, S. T. et al. Quantum capacitance-limited MoS2 biosensors enable remote label-free enzyme measurements. Nanoscale 11, 15622–15632 (2019).
- Guros, N. B. et al. Reproducible Performance Improvements to Monolayer MoS2 Transistors through Exposed Material Forming Gas Annealing. ACS Appl. Mater. Interfaces 11, 16683–16692 (2019).

