Development of a Collagen Fibril Reference Material for Reproducible Cell Response
Summary
An important determinant of cellular response to drugs and toxins is the chemical and physical nature of the extracellular matrix (ECM) to which cells are adhered to within tissues. The cell's ECM environment can drastically affect intracellular pathways, and can alter cell response in various in vitro cell-based assays. Cell-based assays are used extensively in drug discovery, in assessment of toxicity to chemical and biological agents, in research settings to understand intracellular pathway systems, and in some diagnostic and therapeutic settings.
Currently, cellular assays are most often performed in plastic culture dishes which are sometimes modified with ill-defined protein coatings. A standard reference extracellular matrix material that is sufficiently characterized, reproducible, stable, and biologically relevant would improve confidence in the biological relevance of the assay, and in assay reproducibility. Previous results collected at NIST have shown that films composed of Type I collagen fibrils induce reproducible and physiologically relevant cell behaviors similar to those observed on collagen gels, but are easier to handle, are more reproducible, can be rigorously physically characterized, and provide better optical properties for cell imaging.
Description
Research is ongoing to assess what information is required to validate films of collagen as reference extracellular matrix. During the first portion of this work we will develop protocols for characterizing the prototype collagen reference material with various instrumental techniques to develop standard metrics to which different batches can be compared. The second segment of research will focus on determining the reproducibility of cell response on collagen fibrils. Finally, examination of the behavior of cells on fibrils handled in different ways will be used to determine the functional stability of the collagen matrices. Within the first year, and continuing into the second year of funding, we will engage a user community for this material that will provide a test group for assessing the practicality of this material.
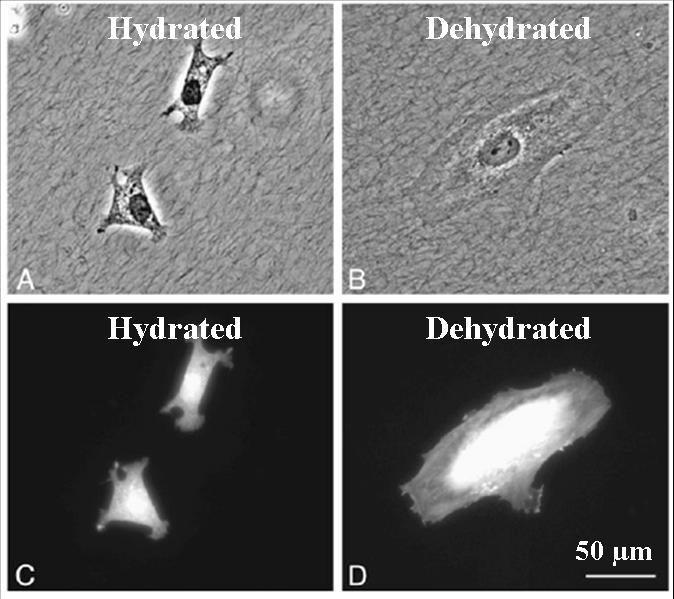
Cellular morphology change on hydrated and dehydrated collagen fibrils, (A,B) phase image, (C,D) cell bodies stained with Tx-Red.
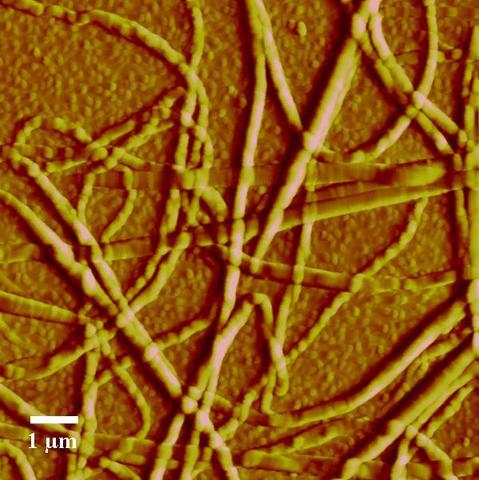
AFM Image of Collagen Fibrils
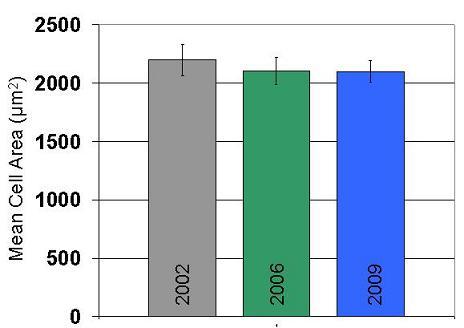
Reproducibility of cell spreading on collagen thin films over several years of study
Major Accomplishments
- Preparation and characterization of robust fibrillar mats of type I collagen on hydrophobic alkanethiol / gold substrates
- Use of collagen films to image cellular interaction with collagen fibrils with live cell microscopy.
- Demonstration of reproducibly of cell response on collagen films over several years
- Developed methods to produce collagen fibril films on untreated polystyrene substrates and PDMS to facilitate usage in conventional multiwall plates.
- Use of collagen films to identify signaling pathways that detect the physical and chemical properties of extracellular matrix.
- Technology used to prepare collagen films has been transferred to several international and national laboratories.
Additional Technical Details
- Elliott, J. T., A. Tona, P. L. Jones, and A. L. Plant. 2002. Highly reproducible fibrillar and non-fibrillar Type I collagen surfaces induce a distribution of responses in smooth muscle cells. Mol. Biol. Cell 13:68A-68A.
- Elliott, J. T., A. Tona, J. T. Woodward, P. L. Jones, and A. L. Plant. 2003. Thin films of collagen affect smooth muscle cell morphology. Langmuir 19:1506-1514.
- Elliott, J. T., J. T. Woodward, K. J. Langenbach, A. Tona, P. L. Jones, and A. L. Plant. 2005. Vascular smooth muscle cell response on thin films of collagen. Matrix Biol. 24:489-502.
- Langenbach, K. J., J. T Elliott, A. Tona, D. McDaniel, and A. L. Plant. 2006. Thin films of Type I collagen for cell by cell analysis of morphology and tenascin-C promoter activity. BMC Biotechnol. 6.
- McDaniel, D. P., G. A. Shaw, J. T. Elliott, K. Bhadriraju, C. Meuse, K. H. Chung, and A. L. Plant. 2007. The stiffness of collagen fibrils influences vascular smooth muscle cell phenotype. Biophys. J. 92:1759-1769.
- Elliott, J. T., M. Halter, A. L. Plant, J. T. Woodward, and A. Tona. 2008. Evaluating the performance of fibrillar collagen films formed at polystyrene surfaces as cell culture substrates. Biointerphases 3:19-28.
- Amyot, F., A. Small, H. Boukari, D. Sackett, J. Elliott, D. McDaniel, A. Plant, and A. Gandjbakhche. 2008. Thin films of oriented collagen fibrils for cell motility studies. Journal of Biomedical Materials Research Part B-Applied Biomaterials 86B:438-443.

