Summary
NIST is building a cold-atom vacuum sensor (CAVS) that will be able to measure pressures in the ultra-high vacuum (UHV) regime corresponding to pressures less than 10-7 Pa or 10-9 torr. Currently, no reliable measurement tools exist at such pressures. The CAVS operates by relating loss of ultracold 1-μK lithium atoms from a conservative trap by collisions with ambient room-temperature atoms and molecules to the back-ground pressure. The dominant background constituent in a UHV is molecular hydrogen, although other gasses can and will be deliberately introduced. My group gives theoretical support for this project by computing a priori elastic cross sections for ultracold lithium with various room-temperature atomic and molecular gasses.
Description
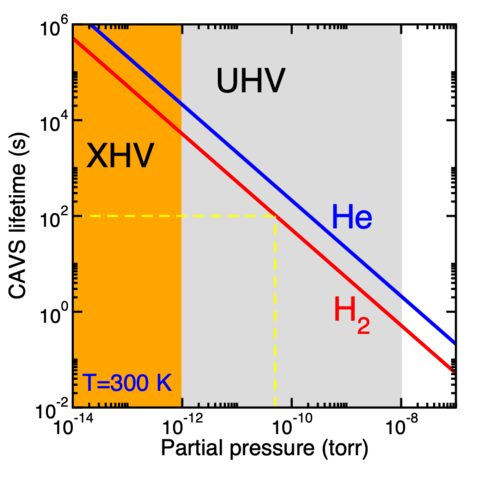
The lifetime of trapped 1-μK lithium atoms in the cold-atom vacuum sensor (CAVS) as a function of partial pressure of 300-K molecular hydrogen (H2) and atomic helium (He) in the UHV domain. The curves are based on our calculations of the elastic rate coefficient between lithium and H2 as well as He.
Measuring pressure in an ultra-high vacuum (UHV) environment is a challenging task. Direct measurement of the force generated by collisions of the residual or background atoms and molecules on a unit area of the vacuum chamber, typically held at or near room temperature, is not feasible. The force is simply too small.
Instead, we envision to insert a small approximately 1 μK sample of the lightest alkali-metal atom, lithium, into the vacuum. The atoms are held in place away from the walls of the chamber by a conservative magnetic trap or bottle, where they have sample sizes that are significantly less than 1 mm. The lithium atoms were cooled to these conditions using laser cooling, a technique pioneered by NIST researchers in the 1980s. The magnetic bottle is engineered to be shallow with trap depths only a few times larger than the mean kinetic energy of the lithium atoms. To good approximation a collision with the nine orders of magnitude hotter residual atoms and molecules will then eject a light lithium atom from the trap. This atom loss leads to an exponential decay of the number of lithium atom with time constant τ that can be accurately measured. An animation of these processes in a cold-atom vacuum sensor (CAVS) can be found here. In principle, the experiment can be setup with either the bosonic or fermionic isotope of lithium.
The next realization is that the time constant for lithium atom loss is proportional to the number density of the atoms and molecules in the UHV with a proportionality constant that is simply the rate coefficient for elastic, momentum changing collisions. The constant is universal as it is the same in any device. It only needs to be measured sufficiently accurately once or be accurately computed from first principle. This property makes the CAVS a primary vacuum pressure standard.
The pressure p then follows from the ideal gas law p=n k T for number density n of residual species at temperature T close to or at room temperature. Here, k is the Boltzmann constant, a constant now exactly defined in the International System of Units (SI). The figure on this page shows the expected lifetimes τ of an ultracold lithium sample in the CAVS as functions of (partial) pressure for T=300 K molecular-hydrogen or helium gasses. Most notable is that for a UHV pressure near 10-10 Torr the expected lifetime is around one minute.
My group theoretically determines the elastic rate coefficients and their temperature dependence for lithium atoms colliding with relevant background atoms and molecules. We pay special attention to assigning accuracies to the rate coefficients. Our aim in the project is to reach relative uncertainties of a few percent. So far, we have determined the elastic rate coefficients and cross sections for molecular hydrogen and helium atoms at the stated accuracy goals. These rate coefficients have been used to construct the figure on this page. Calculations for other species are underway.
The computations have two parts. First, the relevant electronic potentials as functions of the separations between the lithium atom and the atoms in its collision partner are computed with the help of commercially available electronic structure software. Their accuracy is gauged from comparisons of potentials obtained with different basis-set sizes. Second, atoms and molecules are made to rotate and vibrate, i.e. collide, on these potentials with relative kinetic energies up to ten times kT using an in-house coupled-channels software package. Accurate collision rates for hydrogen molecule are only possible because the collision with lithium is non-reactive and the vibrational energy spacing of the hydrogen molecule is an order of larger than kT at room temperature.
The research is performed in collaboration with researchers in the Fundamental Thermodynamics group of the Sensor Science Division.

