Measuring Up: The Phantom of the Library
MRI calibration objects on loan from NIST can dramatically improve measurement quality and value.

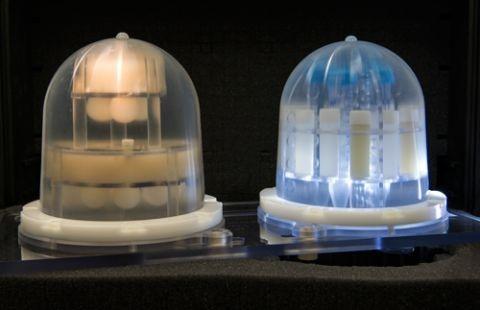
NIST engineer Kathryn Keenan opens an MRI system phantom used for calibrating MRI machines that is traceable to standardized values. The phantom is filled with small plastic containers of various salt solutions that mimic tissues of the human body.
Magnetic resonance imaging (MRI) has revolutionized medical diagnostics and research by providing a non-invasive way to investigate internal tissue structures and processes at millimeter scale, often with far better soft-tissue contrast than can be obtained by x-ray and other methods. MRI is unique in that contrast is dependent on many different tissue properties including water density, water motion, local chemical environment, blood oxygen levels, fat fraction, cellularity, and vascularization.
The value of MRI can be dramatically increased by making its output more quantitative so that images become maps of important tissue properties. Quantitative imaging further ensures that measurements are reproducible, data from one imaging system can be confidently compared to data from another, and performance stability can be monitored over time.
To help achieve those goals, the National Institute of Standards and Technology’s Physical Measurement Laboratory (PML) in collaboration with the National Institute of Biomedical Imaging and Bioengineering (NIBIB) at the National Institutes of Health (NIH) recently launched a unique service that provides MRI research centers and clinics with access to NIST-traceable reference standards.
Nearly all conventional MRI results are qualitative – that is, they show differences in contrast between adjacent areas but do not measure the specific signal properties or intensities that produced those differences. To generate that kind of reliable quantitative data, a scanner has to be calibrated by comparing its measurements of standard reference materials (which mimic various specific tissues or dynamic processes such as fluid diffusion) against the same measurements made by an SI-traceable metrology system.
In medical imaging, such calibration objects are called phantoms. PML’s Magnetic Imaging Group, long a world leader in creating MRI phantoms for quantitative imaging, now offers a “lending library” of phantoms for calibrating whole MRI systems as well as units designed for imaging the breast, heart, knee, and brain. The phantoms contain a wide variety of structures to determine geometric accuracy, image uniformity, and signal-to-noise ratio, along with many different types of tissue mimics that have well-defined properties such as water diffusion, proton spin relaxation, neurochemical composition, and fat content.
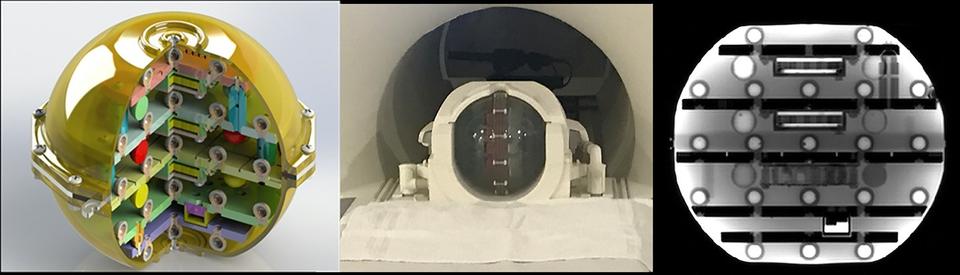
Many of the phantoms in the library were developed by NIST in partnership with organizations such as the National Cancer Institute, the Radiological Society of North America, and the International Society of Magnetic Resonance in Medicine. Others were developed by universities, including the University College London and the University of Michigan, and commercial phantom manufacturers; NIST provided a path for SI-calibration and curation of the phantoms. The phantom lending library is an open resource: users can both check out phantoms and contribute new phantoms to the library.
The lending library provides clinical and research sites, scanner manufacturers, and medical device developers easy access to calibrated phantoms, along with the associated public-domain reference data and analysis software. The phantoms are used in diverse applications such as clinical trials, medical device development, disease research, scanner quality assurance, and scanner monitoring.
“The lending library addresses an important need to have easy access to medical phantoms as well as to have a convenient route to obtain SI-traceable calibration and curation of the phantoms. As image-based in-vivo metrology becomes more complex and widely used, there is a critical need for these measurements to be well-defined and validated,” said Stephen Russek, Project Leader for Imaging Physics.
“We have recently included two new phantoms for brain imaging that assess the accuracy of local measurements of neurotransmitters and scanner stability for functional MRI of brain activity,” he said. “We are looking forward to extending the lending library to include biomimetic phantoms that are inherently multimodal. We will soon be including a triple modality abdominal phantom with realistic kidney and liver lesions that can be imaged with ultrasound, computed tomography, and MRI.”
To date, institutions that have borrowed from the library include: Washington University in St. Louis; University of Illinois; Cornell Medical School; University of California, San Diego; Memorial Sloan Kettering Cancer Center; Case Western Reserve University; University of Southern California; Cleveland Clinic; Boston University; the University of Washington; and Massachusetts General Hospital.
“The lending library is a great complementary service to our biomarker measurement service and they re-enforce each other,” said Quantitative MRI Project scientist Karl Stupic. “As new phantoms are added to the library, the measurement service has to develop methodology to support those phantoms – expanding the capabilities NIST can provide to the MRI community. The lending library has allowed customers to conduct studies across sites and across different systems exploring system variation. These studies have included comparing new MRI systems, different field strengths, and systems undergoing upgrades.”
To obtain MRI images, a patient’s body is placed in a strong magnetic field. The protons (hydrogen nuclei) in the body’s water molecules have a quantum property called spin that makes each proton act somewhat like a bar magnet with north and south poles, which are initially randomly oriented. The applied magnetic field forces the protons’ spins to line up in the same direction.
Then the tissue is irradiated with pulses of radio-frequency waves, which flip the proton spins out of alignment. Between pulses, the protons “relax” to their original aligned states, emitting their own RF waves functioning as a natural nanoscale RFID tag. The imaging system detects and spatially localizes these signals, which have different properties according to the type of tissue and its chemical environment.
To image diffusion of water molecules, MRI uses proton spins as tags on the water molecules and by monitoring spin motion and relaxation, measures the speed at which the water travels. Water diffusion is an important measure of local tissue structure. For example, hypercellular tumors will have restricted water diffusion while necrotic tissue can have enhanced water diffusion. In neural tissue, which is highly anisotropic, water diffusion can measure the direction of neuronal bundles as well as potential disruption caused by traumatic brain injury.
Diffusion imaging is becoming important in breast imaging because it can highlight tumors without the needs for injected contrast agents. “Recent studies of breast cancer patients demonstrated that diffusion can measure response to chemotherapy very quickly, within the first day of treatment,” said Quantitative MRI Project Leader Kathryn Keenan, principal developer of NIST’s breast phantom. “That’s a really exciting development. It is possible to see if a person will respond to the treatment after their first dose. The breast phantom, which is part of the lending library, can be used by an MRI facility to ensure they are making accurate diffusion measurements in the breast coil.”
“Our team had been loaning phantoms to different academic groups and commercial users in an ad hoc manner. I want to thank Stephen for formalizing this effort and NIBIB for helping to fund it,” Keenan said. “We’re excited to see new phantoms contributed and develop an even larger user group.”

Any mention of a commercial product or products is for information purposes only. It does not imply any recommendation or endorsement by NIST.

