
Angela Hight Walker, PML, places a sample for Raman Spectroscopic characterization.
While nanomaterials have led to major advances in key U.S. industries, especially the semiconductor industry, their technological promises and possibilities are tempered by concerns about the possible health and environmental risks associated with their manufacture, use, and disposal.
The health and safety of those involved with the development, manufacture, and use of nanotechnology products is of critical importance. The 2011 National Nanotechnology Initiative (NNI) Environmental, Health, and Safety (EHS) Research Strategy is grounded in the principles of health and safety risk assessment throughout the product life cycle. Working closely within the NNI activity, NIST developed its unique multi-laboratory Nano EHS initiative, which has been funded since 2009 and led by the Materials Measurement Laboratory (MML). Angela Hight Walker, a physicist in the Semiconductor & Dimensional Metrology Division leads the Physical Measurement Laboratory (PML) team that works closely with the NIST lead, Debra Kaiser (MML), and several other principle investigators throughout the campus. A project leader in the Division's newly formed Nanoelectronics Group, Hight Walker's primary focus is to use materials physics to provide the detailed quantitative physical-properties measurements to ensure that risk assessments related to nanomaterials are science based.
"The NIST perspective is that we want to make sure that innovation is spurred with nanotechnology and not hampered by unsubstantiated concerns," Hight Walker explains. "We want to make sure that companies will continue to and even accelerate bringing innovative products that leverage the wonders of nano to market by providing the measurement science to understand and mitigate any potential health and safety risks. Our goal is to provide science-based risk assessment."
The role of Hight Walker's team and other NIST scientists in supporting the Nano EHS initiative is to develop and disseminate measurement tools, based on advances in measurement science, which enable risk assessment and risk management of engineered nanomaterials and nanotechnology-enabled products. The first step of this process is to fully characterize the nanomaterial to assess its physical-chemical properties to precisely identify and quantify what it is. This turns out to be a much a greater challenge than one might think.
When a scientist orders a sample of nanoparticles, for example, carbon nanotubes, some companies that sell them may not have the capabilities to perform a detailed characterization of their product. Impurities in starting materials are common in nanotube samples. This adds a layer of complexity to the characterization process—how can you assess the potential risks of a carbon nanotube when the sample contains impurities that could themselves have health and safety concerns?
"Back in the 2008, the best sample we could get contained about 45 percent nanotubes," Hight Walker states. "While progress has been made, vendors are still challenged in providing samples with purities of 99 percent or better."
Because carbon nanotube samples are inherently "dirty" they must be thoroughly cleaned and impurities separated to ensure that only the purest samples (i.e., those containing a single type of nanotube) are used. This separation is critical to proper risk assessment, and the Hight Walker team relies heavy on the Complex Fluids Group within MML for collaboration in these separations.
"If you want to identify the fundamental properties of nanomaterials, you actually have to isolate the nanomaterial for study," Hight Walker explains. "We want to see what nanotube type is going to give us the desired property and then minimize those that are giving us adverse effects. It really is a combination of separation and characterization."
Hight Walker's team participated in the development of Standard Reference Material 2483, "Single-Wall Carbon Nanotubes (Raw Soot)," which was homogenized and well characterized and released in late 2011. This year, three different lengths of aqueous-based reference material solutions of single-wall nanotubes will be released and available for sale. "We will soon have the necessary reference materials to help people compare measurements on the samples they have with measurements made on NIST validated samples," Hight Walker states.
Hight Walker's characterization efforts have focused primarily on magnetic nanoparticles and carbon nanostructures; nanotubes and graphene rely on optical spectroscopy, especially Raman Spectroscopy. With Raman Spectroscopy, the sample is illuminated with a laser beam. Reflected, inelastically scattered light from the sample is collected with a lens and sent through a monochromator. A detector is able to filter out the unaltered light (Rayleigh scattering) from the Raman scattering. This measurement produces a pattern or signature of a molecule that can be used to help identify it and provide information about physical properties of the material.
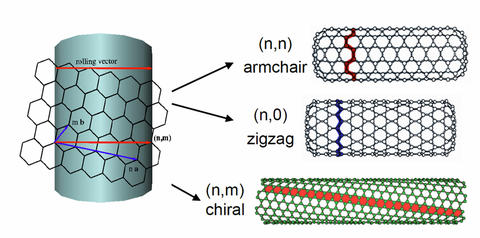
A recent breakthrough, the first demonstrated separation and characterization of pure armchair nanotubes, was again a collaboration between MML and the Hight Walker's group. Armchair nanotubes, the most coveted type of nanotube due to its lack of band gap which allows electrons to flow through without resistance, has long been a dream first discussed by former Rice professor and Nobel prize winner, Richard Smalley, as an ideal quantum wire. [1]
Hight Walker is also the leader of the United States team, US TAG WG2, working within the International Organization for Standardization technical committee ISO TC 229 Nanotechnology to develop internationally accepted standards for nanotechnology. Working closely with industry, academic and regulatory agencies, ISO is heavily involved in both characterization and EHS activities. WG2 has released 9 nanotube measurement protocol standards.
A recent focus for Hight Walker's group is multi-wall carbon nanotubes embedded in polymer matrices. The group is also developing, in collaboration with NIH, toxicity measurements on separated single-wall nanotubes, an area of interest for both nanomedicine and health and safety risk assessment.
"The science is still weak on connecting a single parameter with an actual toxicological response," Hight Walker explains. "The goal is to be able to quantitatively tie specific nanotube properties such as size and dimension to different toxicological outcomes. Also, I do not see in the short term that a single measurement method will be able to provide the level of characterization needed to assess health and safety risk. It is only through interdisciplinary collaborations like the ones we have here at NIST that we can have a large impact on this critical area."
"I care about nanomaterials," Hight Walker concludes. "I want to understand their fundamental properties so that people can make applications that enhance life. Any risk can be managed if it's well understood."

