
The outside of the deposition chamber with researcher Kevin Dwyer looking on, and a close-up of the inside showing the silicon target (glowing orange) where incoming ions will form epitaxial layers.
Any eventual quantum computer, no matter how it may be configured, will need a way to store and manipulate information in qubits – the quantum counterpart of a conventional digital bit, but capable of a coherent "superposition" of multiple values at the same time instead of either 0 or 1. There are many promising possible qubit implementations, including ions, Josephson junctions, optical lattices, quantum dots, electron spin states, and photon polarization. Some are comparatively exotic.
But one appealing option employs the same familiar workhorse metalloid found at the heart of today's computers: silicon, and specifically silicon-28 (28Si), the most abundant isotope. The qubits would be embodied in the spin states of dopant atoms, such as phosphorus, embedded in silicon crystal. Those states can be manipulated by applying radio or microwave frequency pulses, which flip the spin orientation.
As a practical matter, however, such solid-state qubits would have to be placed in 28Si crystal of extraordinary isotopic purity. That's because the key metric for performance of the qubit is the spin decoherence time (T2) – that is, the duration of the coherent superposition state, during which the qubit can be used for quantum information processing. However, silicon's second most common isotope, 29Si (about 4.7% natural abundance), has nuclear properties that interfere with the qubit's integrity. And commercial silicon wafers are only about 92.2% isotopically pure, far too low for workable solid-state qubits.
"Unfortunately," says Joshua Pomeroy of PML's Quantum Measurement Division, "there's only one meaningful channel at present for isotopically enriched 28Si -- the international Avogadro Project, which seeks to make a kilogram standard from an ultra-pure sphere of 28Si. Anybody who's doing anything other than Avogadro, but needs the high purity Si, has to go begging them for pieces. Otherwise, you're out of luck."
But perhaps not for long. Pomeroy's team is developing a method to grow ultra-pure 28Si crystal that could satisfy the needs of the quantum information community at relatively low cost. It begins with a stream of silane (SiH4) from which the hydrogen is cracked and the silicon atoms are ionized and accelerated at about 4 kV through a magnetic field that bends the atoms' trajectories differently according to mass and charge, as in a mass spectrometer, spatially separating the three isotopes of silicon in the beam. (See Figure 1.) The 28Si ions are then routed into a high-vacuum chamber in which they are focused and deposited onto a natural silicon substrate about 4 mm wide.
"From that point on" Pomeroy says, "it's essentially molecular-beam epitaxy (MBE). We want to make what's known as epitaxial layers – that is, single-crystal layers that are lattice-matched to the substrate. For the substrate, we start with a piece of natural-abundance silicon wafer which we will need to heat to about 1250 °C so that it's atomically clean on the surface. Our expectation, based on other work, is that we would only need to produce 100 nm or 200 nm of material for it to look to the quantum system just as though the entire wafer was enriched. So we only need to produce a microgram as opposed to a kilogram that they need to provide for the Avogadro project."
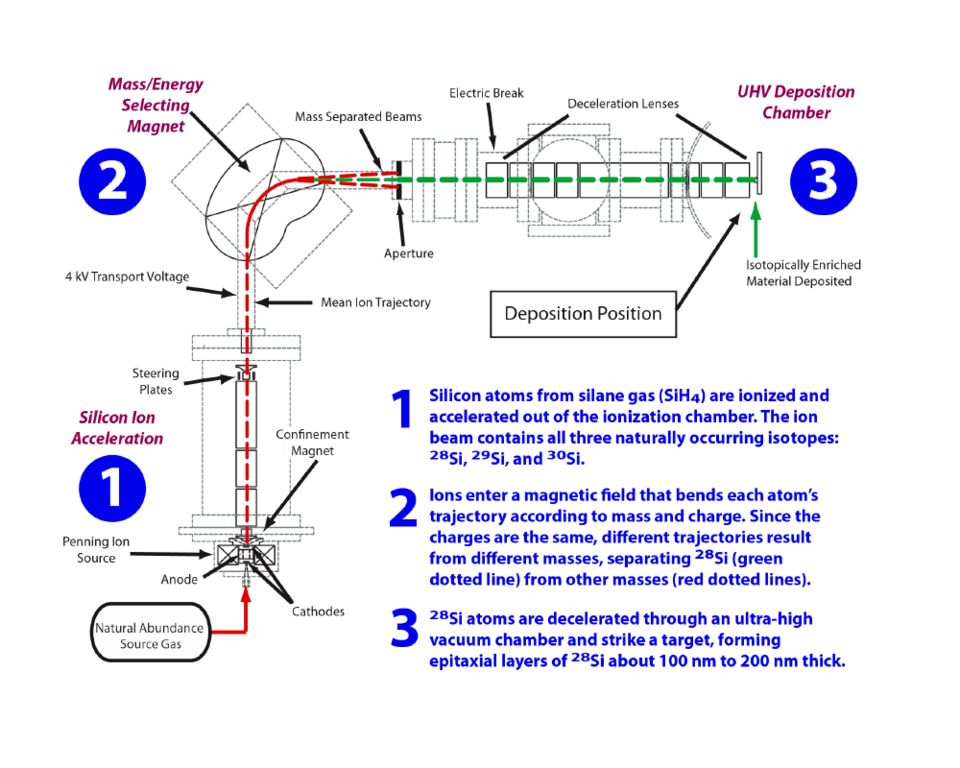
How pure is pure enough? There is no specific value, Pomeroy says, "but at the end of the day, what really matters is the T2 time. In general, researchers think that for every order of magnitude improvement in reducing the 29Si, you get an order of magnitude improvement in the T2 time. In fact, for prime-grade commercial silicon, the single limiting factor in T2 time is the 29Si density."
The project's goal is to achieve "four nines" (99.99% isotopic purity), and earlier this year Pomeroy and colleagues succeeded in reaching 99.9% 28Si. It took two years to get to that point, in a staged process that began with tests on purifying neon and then moved to isolation of 12C. With carbon, the team achieved 99.996% 12C enrichment, as determined by secondary ion mass spectrometry, which measures the chemical identity and relative abundance of ions displaced from a sample surface.
"To my knowledge, we're the only group in the western hemisphere doing in situ enrichment and deposition, and with this particular method we may be the only group in the world," Pomeroy says. "And now that we've enriched silicon by a factor of 80, some other labs and programs are interested."
Getting to the next order-of-magnitude improvement will take some time. Pomeroy says that the present concentration of 29Si on the substrate probably results from background gas contamination at the sample. "So whatever our ultimate enrichment is," Pomeroy says, "it's probably not going to be limited by mass selectivity, but by the background gas problem. There are two things we need to do to address that problem.
"One is getting to ultra-high vacuum in the epitaxy chamber, around 10-10 Torr. Currently we've been using a gas pressure for the silicon about four orders of magnitude higher than when we did the 12C experiments. The second is to increase the 28Si beam fluence because we can outrun the effect of background gas contamination by growing our layers faster. Right now, our growth rate is much lower than it should be, and we've been tearing the machine apart to find out where our missing ion current is."
Pomeroy thinks that the project could easily take five to 10 years to reach fruition. While the main focus thus far has been improving the enrichment, ultimately the new silicon must also be ultra-pure chemically and have crystalline perfection similar to conventional microelectronics silicon. In the long run, assuming those criteria are met, "our aim is not to become a foundry for this material," Pomeroy says, "but to have demonstrated a process that could become a sort of quarter-million-dollar solution that anybody could install. It would be just like adding another MBE tool to your lab, and you wouldn't have to worry about getting into a high-risk venture."
If successful, the project has the potential to reduce the cost of entry for mainstream industry to use isotopically enriched silicon as part of thermal management solutions or for development of quantum information systems.

