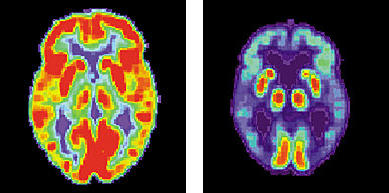
Two examples of positron emission tomography (PET) scans. The left image shows a normal brain, the right shows a brain with characteristics of Alzheimer's disease.
Broadway stars have understudies. Now, an increasingly popular radioactive isotope has its own stand-in. Developed in part by researchers at the National Institute of Standards and Technology (NIST), the substance might ultimately improve medical imaging, speed up clinical trials of many drugs and facilitate efforts to develop more individualized medical treatment.
The number of medical images obtained through the technique known as positron emission tomography (PET) is increasing at a rate of 20 percent a year—and this has correspondingly increased the use of fluorine-18 (18F), the radioactive isotope of choice in the vast majority of PET procedures. Injected into the bloodstream while bound to "carrier" molecules, fluorine-18 lights up the body during PET scans to perform such jobs as revealing tumors, monitoring heart activity and determining which regions of the brain are active during certain tasks.
However, fluorine-18 has a very short life—a batch of the stuff decays to half its initial quantity in fewer than two hours, and less than one-ten-thousandth of the original amount is left after a single day. Because the stuff decays so quickly, it is not possible for a single central lab to make precise measurements on fluorine-18 solutions and then distribute them to far-flung centers to calibrate PET machines. This lack of a "standard reference" for fluorine-18 means that PET-related measurements from center to center, from patient to patient, and even in the same patient over time, are difficult or impossible to compare with one another.
NIST researchers, collaborating with an Ohio-based nuclear medicine company called RadQual, have turned to germanium-68 (68Ge) as a calibration surrogate for fluorine-18. This germanium isotope is much longer lived—its half-life is 270.95 days—but its radiation decay characteristics are otherwise similar to fluorine-18. Small differences between measurements of germanium-68 and fluorine-18 can be accounted for through the use of mathematical "conversion factors." Germanium-68 never would be used in actual PET procedures; it would only be used to calibrate the machines properly, enabling more accurate measurements of the fluorine-18 isotopes. Although germanium-68 has been used for performing corrections on PET scanners, the industry has lacked a nationally traceable standard reference material for instrument calibration.
NIST researchers have calibrated solutions of germanium-68 that could become the basis of a new standard reference material for this isotope. The next phase of this project involves working with RadQual to calibrate a new "mock" syringe standard that would use germanium-68 embedded in an epoxy to simulate fluorine-18 in a syringe. This would help researchers more accurately determine the amount of fluorine-18 to be injected into patients during the PET procedure so as to minimize radiation dose while still producing the best image. NIST is also working independently on a 68Ge-based PET "phantom" that can be used to calibrate PET scanners in a way that is traceable back to NIST.
The first commercial products from this collaboration may be reaching PET centers later this year. By giving doctors more accurate results on how specific patients are responding to cancer treatments and providing more precise indications earlier in clinical trials as to how individual patients are metabolizing particular drugs, better standards for PET imaging may hasten the arrival of "individualized" medicine.

