HP-BioSANS (LIPSS)
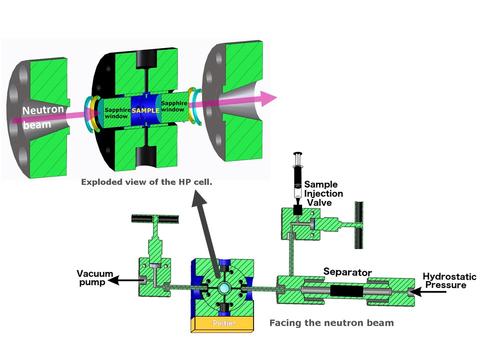
High-Pressure BioSANS
LIQUID INSERTION PRESSURE SYSTEM FOR SANS (LIPSS)
LIPSS enables in situ SANS measurements under broad ranges of pressure and temperature, so that sample environment effects can be probed from minutes to hours1. No radiation damage and an extended q-range make HP-SANS complementary to HP-SAXS measurements, providing information on a broad range of molecular sizes, shape, aggregation, folding/unfolding, etc. The non destructive nature of neutrons allows for hysteresis effects to be probed, while contrast variation SANS can be used to highlight contributions to scattering from specific components of complex macromolecular assemblies such as viruses2 or others.
LIPSS is a high-pressure (HP) system (see specifications below): the McHugh sample cell is made of a high-nickel-content austenitic steel and is capable of withstanding a maximum pressure up to 350 MPa (3.5 kbar). A Peltier system controls the temperature between -20°C and +65°C: under pressure, subzero temperatures can be accessed in the absence of ice. The sample is loaded through an injection system that minimizes air bubbles and fills the cell completely. A separator isolates the sample from the pressurizing medium, as shown schematically in the image1. LIPSS was designed for low viscosity solutions and its performance depends on the specific environment required. It is strongly recommended that you discuss measurement needs in advance, with Susana Teixeira [scm5(at)nist.gov] or your local contact.
Past and potential applications of HP-SANS and the LIPSS sample environment include studies on:
- cold denaturation of proteins3
- storage conditions and stability of monoclonal antibodies4-6
- high-pressure low-temperature processing of food proteins7
- high-pressure response of amyloid folds8
- self-assembled lipid nanoparticles9
- viral inactivation10
- pressure-assisted enzymatic activity11
- protein aggregation under pressure12,13
- lipid phase transitions14
- surfactant self-assembly at high pressure15
- polymer blend nucleation and interactions16,17
- engineering baroplastic behavior of block copolymers18
- adaptations to extreme environment: life under pressure19
- combined effects of salts and high hydrostatic-pressure20
Let us know about the sample environment you need
There are ongoing efforts at the NCNR to optimize the HP BioSANS system described above, namely through improved automation, thermal insulation and improved cooling/heating power. Contact Susana Teixeira to find out more, or discuss the specific needs of your experiment.
REFERENCES
- Teixeira et al., High Pressure Cell for Bio-SANS Studies Under Sub-zero Temperatures or Heat Denaturing Conditions’. J. Neutron Res. 20,13 – 23 (2018).
- He et al., Conformational changes in Sindbis virus induced by decreased pH are revealed by small-angle neutron scattering. J. Virol. 86, 1982-1987 (2012)
- Dias et al., The hydrophobic effect and its role in cold denaturation, Cryobiology 60 ,91–99 (2010).
- Berger et al., High Pressure Light Scattering of therapeutic proteins to probe Aggregation and Protein-Protein Interactions.. J. Chem. Phys. B 127, 5742- 5754 (2023).
- Gomet et al., In Situ Monitoring of Protein Unfolding/Structural States under Cold High-Pressure Stress. Molecular Pharm., 10.1021/acs.molpharmaceut.1c00604 (2021).
- Berger et al., High-Pressure, Low-Temperature Induced Unfolding and Aggregation of Monoclonal Antibodies: Role of the Fc and Fab Fragments, J. Phys. Chem. B., doi: 10.1021/acs.jpcb.1c10528 (2022).
- S.C.M Teixeira. High-pressure small-angle neutron scattering for food studies, Curr. Op. Colloid & Interface Science 42, 99-109 (2019).
- Torrent et al., High pressure response of amyloid folds. Viruses 11(3): 202 (2019).
- Kulkarni et al., Effects of High Pressure on Internally Self-Assembled Lipid Nanoparticles: A Synchrotron Small-Angle X-ray Scattering (SAXS) Study. Langmuir 32, 45, 11907–11917 (2016).
-
Oliveira et al., Low Temperature and Pressure Stability of Picornaviruses: Implications for Virus Uncoating. Biophysical J. 76, 3, 1270-1279 (1999).
-
Habinshuti et al., Recent and novel processing technologies coupled with enzymatic hydrolysis to enhance the production of antioxidant peptides from food proteins: A review. Food Chem. 423, 136313 (2023).
-
Jackson & McGillivray. Protein aggregate structure under high pressure. Chem. Commun. 47, 487–489 (2011).
-
Seefeldt et al., High-pressure studies of aggregation of recombinant human interleukin-1 receptor antagonist: Thermodynamics, kinetics, and application to accelerated formulation studies. Protein Science 14(9): 2258–2266 (2005).
-
Hammouda and Clover. SANS from P85/Water-d under Pressure. Langmuir 26(9), 6625–6629 (2010).
-
Leseman et al., Self-Assembly at High Pressures: SANS Study of the Effect of Pressure on Microstructure of C8E5 Micelles in Water. Ind. Eng. Chem. Res. 42, 6425-6430 (2003).
-
Patel et al., Observing Nucleation Close to the Binodal by Perturbing Metastable Polymer Blends. Macromol. 40(5), 1675 (2007).
-
Ruegg et al., Effect of Pressure on a Multicomponent A/B/A-C Polymer Blend with Attractive and Repulsive Interactions. Macromol. 40(2), 355 (2007).
-
Ruzette et al., Pressure effects on the phase behavior of styrene/n-alkyl methacrylate block copolymers. Macromol. 36(9), 3351 (2003).
-
Penhalurick et al., Pressure-effects and adaptation mechanisms of ambient and deep-sea bacterial enzymes. Biophys. J. 116(3), 342a (2019).
-
Paul et al., Probing pressure-driven protein phase behavior via in situ high-pressure scattering methods. Acta Cryst. A 79, a127 (2022).
USEFUL LINKS
- McHugh Cell assembly instructions
- BL2 Biomolecular Labeling Laboratory: supports labeling proteins and nucleic acids with 2H, 13C and 15N. It also has the capability for domain labeling and amino acid-specific labeling of proteins expressed in bacteria and yeast.
- SASSIE program suite to generate and manipulate large numbers of structures and to calculate the SANS, SAXS, and neutron reflectivity profiles from atomistic structures. Includes a Biomolecular Scattering Length density calculator.
- Sasview: small angle scattering data analysis software package
- NSF-funded Research Coordination Network (RCN) on Extreme Biophysics
-
High Throughput, High-Pressure Small-Angle Neutron Scattering Sample Environment: a prototype work-horse system designed for low temperature measurements.
DISCLAIMER
Certain trade names and company products are mentioned in the text or identified. In no case does such identification imply recommendation or endorsement by the National Institute of Standards and Technology (NIST), nor does it imply that the products are necessarily the best available for the purpose.
By selecting almost any of these links, you will be leaving NIST webspace. We provide these links because they may have information of interest to you. No inferences should be drawn because some sites are referenced, or not, from this page. There may be other web sites that are more appropriate for your purpose. NIST does not necessarily endorse the views expressed, or concur with the assertions presented on these sites. Further, NIST does not endorse any commercial products that may be mentioned on these sites.
Specifications/Capabilities
- Pressure range (MPa): 0.1 - 350 (ambient to 3.5 kbar)
- Temperature range (°C): -20 to +65 (higher temperatures possible§)
- Sample volume (mL): 2 to 5
- Typical sample concentration: 5 mg/mL or higher (depends on contrast§)
- Can I recover my sample? Yes, typical recovery of ~2/3 of the volume
- Sample thickness (mm): 1 to 5 (neutron path length across the sample solution)
- Q range (Å-1): 0.003 to 0.3 (SANS); 0.0001 to 0.003 (USANS BT5 instrument)
- Type of samples: Liquid (solutions or stable suspensions)
- Real space distances: ~30 Å up to ~6 µm (depends on resolution and contrast§)
- Training required? Yes, please contact S.Teixeira or J.Leão to arrange for training in advance.
Compatible with NCNR instruments: NGB30SANS, USANS (BT5), NG7SANS, 10m SANS.
§ Please contact Susana Teixeira if you have questions regarding feasibility.

