Taking Measure
Just a Standard Blog
As a happily retired materials scientist from NIST, I have continued a long-standing interest in how materials deform and fracture under high stress. My research began in the 1960s with fundamental investigations into the way intrinsically brittle solids can sustain highly concentrated stresses beneath hard contacts. (An everyday example of such a contact is a round pellet fired from a BB gun at a windowpane.) Later, my focus turned to studying materials in a broad range of small-scale technological applications, such as semiconductor chips, electronic sensors, optical lenses and multilayered films. In these small-scale devices, contact with a single errant particle on an otherwise pristine surface can reduce mechanical strength by several hundreds of times, which in turn can lead to premature catastrophic device failure.
Why Teeth?
I became interested in teeth late in my career, initially as a sideline to my physical science research interests. It all began when dental researchers from New York University (NYU) approached me. They sought to understand the basic mechanisms of tooth breakdown, so that practitioners might adapt superior methodologies to prolong the lifetime of our teeth and their restorations. Dentistry is a multibillion-dollar industry, so even the smallest technological advances can have vast financial impact. Those NYU researchers had both dental and engineering training, an unusual combination but one that facilitated an uncommon understanding of the underlying factors governing tooth survival. They recognized that biting is essentially a tooth-to-tooth contact process, and that understanding the mechanisms of tooth damage required input from materials science. My sideline interest soon developed into a larger-scale program, with a network of internal and external collaborators and with multimillion dollar funding from the dental branch of the National Institutes of Health (NIH).
Teeth, like bones, are brittle, yet have to provide structural support to our repetitive mouth motion over a lifetime of food intake. It turns out that tooth enamel is full of structural defects, “tuft”-like microcracks at the interface with the underlying softer dentin. Longitudinal fractures initiate from these tufts and extend vertically along the outer walls of the enamel shell. Such defects and cracks are present in all our teeth and proliferate with age. Yet teeth retain a remarkable inbuilt resilience — they are “flaw-tolerant.” What drives dental research is the need to develop new and better restorative materials that can withstand high bite forces, and in this endeavor the work coming out of NIST has helped produce some dramatic improvements. Go to a dentist now for a prosthetic crown or bridge and you will get a strong ceramic replacement with less toxicity than their old metal-based counterparts and with superior aesthetics. That advance is due solely to the input from materials scientists.
Dentists tend to be practical folk, more technician than scientist. They may adopt the latest and greatest restorative materials, but often have scant interest in the underlying mechanics of how teeth and their replacements actually support load. Like many in the medical field, their modus operandi centers around the culture of the clinical trial — try it out and see how long it lasts. It was only when I subsequently made contact with biologists that a better understanding of tooth function became apparent. Paleontologists in particular have a great interest in teeth, first because teeth make up some 90% of the fossil record and second because crack patterns on tooth surfaces are a fingerprint of dietary history. Accordingly, the study of “dental fracture mechanics” has blossomed into a cottage industry in evolutionary biology.
Mechanisms of Tooth Survival
So what actually contributes to the remarkable resilience of teeth? How can brittle enamel sustain multiple cracks without failing? As mentioned above, enamel contains intrinsic defects that originate during the early stages of development, and which provide starter sources for crack initiation. Part of the explanation lies in the rounded topography of the tooth cusp, notably in the molars. The bulk of the tooth bite force is transmitted as a compressive stress down the walls of the enamel shell — much like the way the dome on a cathedral transmits its weight to the structural base. This compressive stress suppresses crack growth. Another part resides in the enamel’s hierarchical structure (structure within structure) with mineralized rods or prisms bound by protein sheaths. These rods become intertwined into a basket-like pattern adjacent to the dentin junction, impeding any intrusive cracks from penetrating into the vulnerable interior. Teeth are built to last.
By way of demonstration, we conducted failure tests on molar teeth from human patients (provided by the American Dental Association) and some expired sea otters (provided by the California Department of Fish and Wildlife’s Marine Wildlife Veterinary Care and Research Center). We also ran dummy tests on specimens consisting of glass shells infilled with resin, simulating enamel on dentin. These latter tests provided a simplistic but valuable method for elucidating how tooth damage can develop while chewing, and for establishing the essential mechanics that govern tooth integrity. Cracks could be driven down the side walls, but remain highly stable, meaning that they extend incrementally with each small increase in bite force — it is only in a spurious force overload that such cracks are driven to full failure. In our experiments, some cracks originated at the top surface, others from the tooth margin, depending on whether the contact was applied with a “hard” or “soft” indenting object. Critically, distinguishing between these two crack origins helps biologists determine the nature of diets for different animals, specifically whether they tend to eat hard or soft foods.
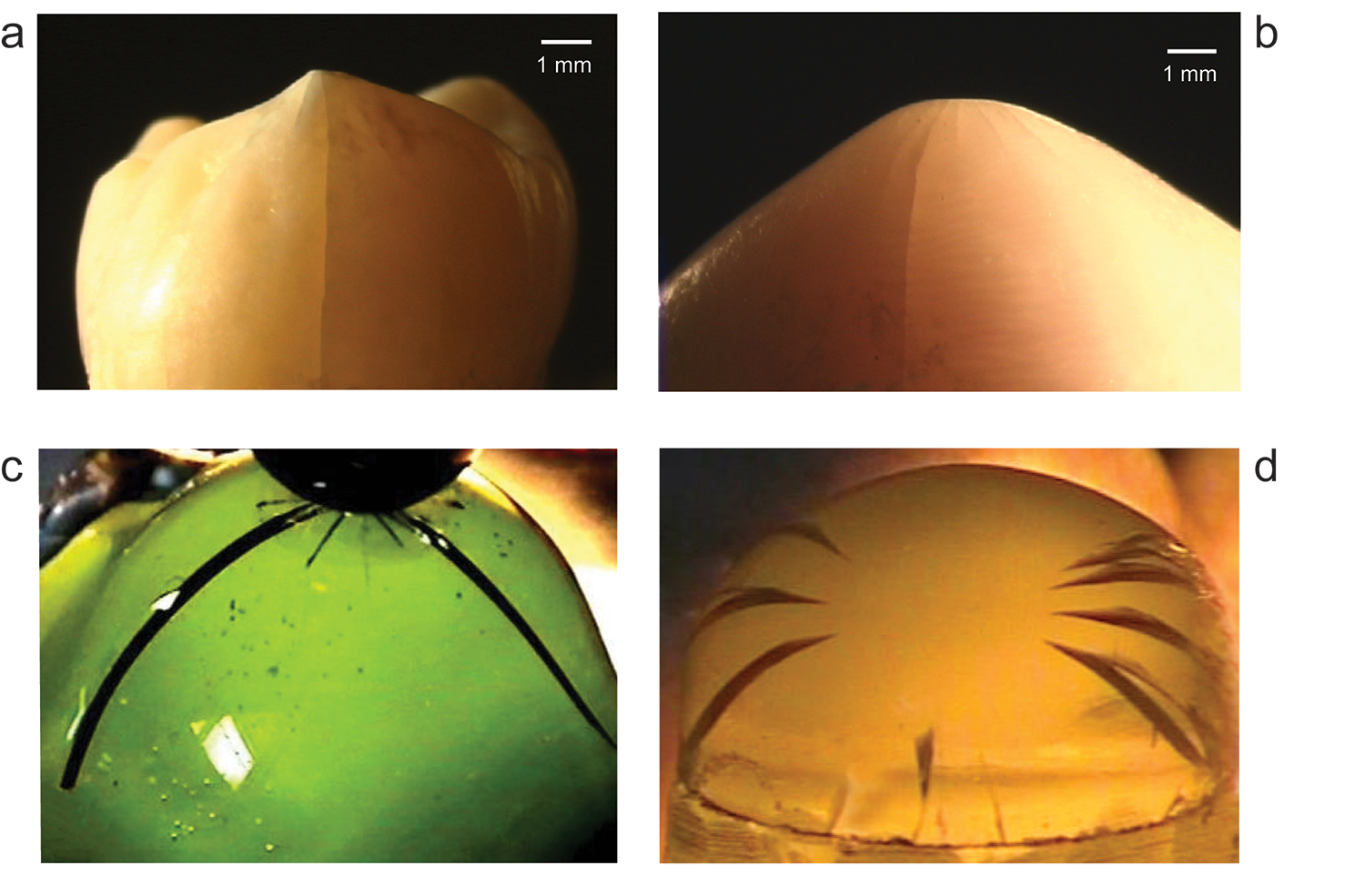
Tooth fracture patterns vary dramatically from species to species — omnivores, carnivores, herbivores. We studied various fracture modes in these different species, shedding light on evolutionary pathways. “Show me your teeth, and I will tell you who you are,” said Georges Cuvier, a celebrated 18th-century scientist. We also developed analytical equations relating critical bite force (the force at which tooth failure occurs) to important tooth parameters, such as tooth size and enamel thickness. Those relations enable biologists to estimate critical bite forces for any given species from tooth geometry alone, whereas previously such determinations required time-consuming, elaborate jaw mechanics analysis.
So, why should we care? Well, quite apart from providing clues as to how we got here, unlocking the secrets of nature is leading to an expansion in biomimicry, in which simulation of natural biological materials provides a whole new route to the design and development of new composite and multilayer structures that have greater resistance to failure, and that are finding new places in manufacturing in bio- and nano-engineering.
Epilogue
An aside concerning dentists and biologists: TV science programs often feature evolutionary paleontologists, rarely dentists. Two of my postdocs took different career paths into academia, one in a dental school and the other in a biology department, where they are both now full professors. The starting salary for the dental researcher was more than double that of the biologist. Sometimes we have to decide on conflicting aspirations: fame or fortune. For those of us who stick with a federal lab it is likely neither of the above — but rather one of dedicated service to the love of science. Science is an international endeavor, and I remain especially appreciative of the NIST guest scientist program, which prior to my retirement enabled researchers and students from all over the globe to work with me in my lab. It taught me that science knows no international boundaries, and that we all have essentially the same DNA.
About the author
Related Posts
Comments
Excellent article. I love the part where you state that dentist are more technicians than investigators. How true. That is why I think we should design and create a new career as Dental Engineering to promote the more scientific approach of an actual mediocre career.
looking for why teeth stain yellow





You have a good point here!I totally agree with what you have said!!Thanks for sharing your views...hope more people will read this article!!!