Taking Measure
Just a Standard Blog
My fascination with the microbial world began when I was around 7 years old and my mother bought me a book called “The Value of Believing in Yourself: The Story of Louis Pasteur”. Nearly four decades later, I still vividly remember the story about the little boy Joey and the “invisible enemy” that had invaded his body.
So began my journey into this tiny realm.
On Friday, May 13, the White House announced a new initiative to advance our knowledge of the world of microbes, called microbiomes, and realize their full potential. As it has for so many other innovations, NIST will play an important part in the realization of microbiome-based products and treatments by helping to develop standards and measurement tools that will foster innovation and confidence for stakeholders in biomedicine and healthcare, biomanufacturing, food safety and environmental health.
I’m pretty excited.

Before I came to NIST in 2014, I worked for 11 years as a geneticist at the Food and Drug Administration (FDA) identifying the particular strains of bacteria that had caused food-borne outbreaks of illness. For example, if people were getting sick from eating pre-bagged leaf spinach contaminated with E. coli O157:H7, we had DNA-based molecular tools to show how that strain of E. coli was unique and different from strains implicated in previous outbreaks. That information helped public health investigators identify outbreak “clusters” and even the food processing plants or the farmers’ fields where the contaminated food had originated. Today, we call this approach “molecular epidemiology.”
It’s like CSI, but the suspect is a microbe.
Molecular epidemiologists “read” the DNA of bacteria to identify them. Twenty years ago, it might take years and cost millions of dollars to read all the DNA (the genome) in a single bacterium. Over the past decade, however, advances in DNA sequencing technology have made genome sequencing faster and less expensive. Today, we can sequence the entire genome of a single bacterium in about a day for about $100.
But a single bacterium? Boring.
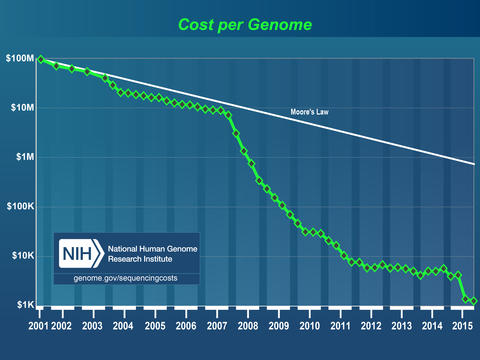
With prices plummeting, many molecular geneticists like me have—with kid-like curiosity and delight—begun shoving heaps of gobbledygook (that’s a technical term for soil, poop, spit, produce, the contents of vacuum cleaners, belly button lint, etc.) into our DNA sequencing instruments just to marvel at what comes out the other side.
What we’ve found is that there’s even more bacteria, and more kinds of bacteria, around us than we ever imagined. There are bacteria literally everywhere on Earth: in the soil, oceans, air (you’re breathing in bacteria right now), on our skin and in our guts. The diverse bacteria that live in-and-on our bodies are collectively referred to as the “human microbiome.”
If we’re talking numbers of cells in the human body, there are far more of them than there are of us.
The good news for germophobes is that we live in harmony with these bacteria. In fact, human and environmental health depends on communities of “good” bacteria. This realization represents a paradigm shift in the world of microbiology. From about the mid-1800s until just recently, scientists thought that these little critters only caused disease and other bad things like food spoilage. As we’ve started to learn more about these microbiomes and how they influence living systems and the environment, we have begun to imagine harnessing them for all kinds of applications, such as gobbling up pollutants, making biofuels, fertilizing soil, improving human health and treating disease.
The First Leg of the Stool (pun intended): Therapeutics
Personally, I’m most fascinated by the therapeutic potential of microbiomes. Bugs as drugs! Already, fecal transplants have been used to successfully treat infections of C. difficile, a potentially deadly bacterium that is usually dormant in our intestines but can proliferate after antibiotic treatments kill the “good” bacteria that keep it in check. Transplants of feces (the gut microbiome) from healthy volunteers via a colonoscopy or other methods (search for that on YouTube at your own risk!) have a cure rate as high as 95 percent! I’ve heard medical doctors explain that a cure rate of 95 percent is the highest they’ve seen of ANY therapeutic.
Perhaps what’s most motivating about this successful, albeit unusual, treatment is the realization that “good” bacteria can be used to cure infectious diseases caused by “bad” bacteria without having to resort to using antibiotics. Developing new approaches to treating disease like this are especially important when you consider the growing world health crisis of antibiotic resistance.
But it’s not just about fighting infectious diseases. In the past 10 years, the human microbiome has been implicated in a wide variety of serious health conditions including obesity, mental illness, cancer, and a plethora of autoimmune disorders. We are at the beginning of a new era of medicine where we will be able to harness bacteria to treat a broad spectrum of diseases.
This isn’t science fiction. A handful of pharmaceutical companies are already developing microbiome-based therapeutics. Not the probiotics you get over the counter, but rather, treatments that are tested and verified to be effective at curing a real disease through human clinical trials.
(And I don’t mean to pooh-pooh probiotics. I take a probiotic every morning.)
Putting my FDA hat back on for a moment: In order for the FDA to approve these new treatments for clinical use, they have to satisfy a pretty strict set of criteria for demonstrating their purity, identity, stability, safety and efficacy. As someone trained in chemistry, microbiology and molecular genetics, this is something that keeps me up at night (and I’m sure many a pharmaceutical company head of drug development). Namely, if I have a mixture of bacteria in a pill form and someone asks me to demonstrate its purity, what does the purity of a mixture of bacteria even mean? Is it genetic purity at the species or strain level? Do spontaneous mutations in the genomes count as impurities? Maybe it’s chemical or metabolite purity? Or is it purity relative to outside contaminants like other bacteria that aren’t supposed to be there?
On that note, in a complex mixture, who’s really supposed to be there? Who isn’t?
These things are going to have to be figured out and (re)defined for emerging microbial therapeutics. New measurements will have to be developed and validated. This is where NIST earns its keep, we partner with industry and help move them down the path toward new and improved clinical products.
The Second Leg of the Stool (pun intended): Diagnostics
Our microbiomes might help us diagnose disease and other health disorders, too. Most of our microbiome lives in our gut. In the very near future, you might visit a clinic and give a fecal sample instead of blood or urine. A lab would sequence DNA from the stool to identify your gut microbes. If they found you had an unhealthy microbiome, your doctor could order an intervention strategy such as a change in diet or even a tailored microbial cocktail designed to “reset” your microbiome to a healthy state. These diagnostic techniques could catch disease way before you begin to feel unwell; perhaps even when you’re a baby. Moreover, such personalized treatment strategies serve as the basis for the burgeoning field of precision medicine that promises to kick our ability to fight disease up to notches unknown over the coming decades.
But we’re not quite there. The major challenge in gut microbiome measurements is that every gut is made up of a mixture of hundreds of different species of bacteria, and everyone’s gut is vastly different. So it’s hard for us to know which bacteria are playing an important role, which are stowaways, and which are contaminants. And what about their relative abundance? What defines “healthy”? Is more or less of any one species important for health and disease?
These questions become even more difficult to answer when my collaborator down the street and I can’t even agree on which bacteria are present or their relative abundance in a given sample.
This uncertainty arises because different labs analyze microbiome samples using different protocols and equipment. It’s sort of a personal preference thing—use what you feel comfortable with. If I gave two different laboratories half of the same stool sample for the same kind of analysis, I would likely get strikingly different answers from each lab. How can we use microbiomes to diagnose disease if every lab that runs the diagnostic test gives us a different answer?
So even before we get to the interpretation stage, microbiome researchers need standard collection, storage, measurement, and analysis protocols that will help ensure that their results can be shared and compared across laboratories. They need reference materials—shared samples of a microbiome that are homogeneous and stable and have been exhaustively characterized—to help them assess their performance and identify and understand biases that crop up throughout the measurement process from sample collection to DNA extraction to sequencing. And there needs to be critically evaluated standard reference data so that we can evaluate bioinformatic analysis pipelines and compare them for accuracy and sensitivity.
The Third Leg of the Stool (pun … ok, that’s getting old): Answering the Call
A recent paper published in Nature Microbiology identified the need for the development of standards that will make it easier to translate microbiome research into treatments and diagnostic tools. This is another area where NIST can have an impact. NIST has already succeeded in similar efforts like the Genome in a Bottle Consortium, which resulted in the world’s first human genome reference material. This summer, my colleagues from NIST and the National Institutes of Health will assemble representatives from government, academia, and the biotech industry to kick off a similar effort at the Standards for Microbiome Measurements Workshop. There we will discuss microbiome-specific measurement needs and prioritize ways that NIST can help make their work successful.
This is an exciting time to be working in the life sciences. We are facing enormous challenges, but our little friends have presented us with an enormous opportunity. We just need to get to know them a little better. We need to develop a common language and techniques to describe and evaluate them before we can really put them to work for us. That’s the job my colleagues and I have ahead of us. I would never have thought when I was a boy reading about Louis Pasteur that I’d be an adult about to be up to his elbows in gut bacteria, but the potential they have for improving human health has me anxious to get my hands dirty.
Follow the conversation on Twitter using #NISTMicrobiome. As part of its commitment to the White House National Microbiome Initiative, the NIST-NIH Standards for Microbiome Measurements workshop, to be held Aug. 9-10, 2016, will identify and plan for the development of new reference materials, protocols, and measurement tools to enable the translation of microbiome science into clinical applications.
About the author
Related Posts
Comments
- Reply





