Taking Measure
Just a Standard Blog

On April 13, 1975, in the community of Wheaton, Maryland, a lone shooter allegedly targeting Black people wounded five and took the lives of two others over the course of 30 minutes before he was killed by police.
The two slain victims, Connie Stanley and John Sligh Jr., were both chemists at NIST (known at the time as the National Bureau of Standards). News reports indicated that they might have known each other through their work.
But they were in separate cars that evening and happened to be in that location for wholly different reasons. Connie was driving from a Sunday family dinner. John and his wife (who also worked at NIST, in procurement) had just gone to see The Godfather: Part II in the movie theater at the nearby Wheaton Plaza mall.
This blog post is not dedicated to the event itself, but to the stories of the NIST family we lost and their futures in science, which were cut short.
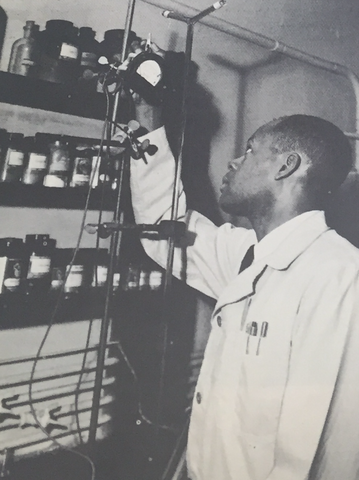
Connie Stanley

South Carolina born and raised, Connie earned his bachelor’s degree in chemistry at South Carolina State College in 1949. He then traveled to Washington, D.C., where he earned a master’s degree from Howard University in 1953 and became part of the NIST workforce one year later.
When he started his career at NIST, Connie turned to titanium. This metal had a high strength-to-weight ratio that made it a competitor with steel and aluminum for structural purposes, but it was more susceptible to wear and also oxidized at higher temperatures, which could lead to corrosion. Previous attempts to minimize these issues by coating the titanium in an oxidation-resistant metal hadn’t been completely successful, but that's where Connie came in. As part of a project for the Defense Department's Springfield Armory, he successfully laid down a protective coating on titanium, which meant the possibility of using the metal at higher temperatures (useful in car engines or gas turbines, for example).
In the late 1950s and 1960s, Connie pivoted to preparing and determining physical properties of pure hydrocarbons. This was part of a larger challenge of accurately measuring the properties of kerosene-like fuels, including combustion temperature. He also synthesized and purified organic compounds for use in “classified research,” as he described in the letter shown here.

NIST historical accounts indicate that, while in the Office of Standard Reference Materials (SRMs) in the early 1970s, Connie also participated in the development of an SRM for cholesterol purity. An iteration of the SRM is still produced today and used by clinical labs for quality control.
Outside of his scientific career, Connie was a member of the Kappa Alpha Psi Fraternity, a scout leader and a member of the Peoples Congregational Church, and he was in a bowling league. He is survived by two children, three grandchildren and one great-grandchild.
Karen Williams, Connie’s daughter, writes: “As a Black man in the scientific community, we are quite sure he overcame many obstacles both professionally and personally, as we all have since his passing.”
John Sligh Jr.

Also originally from South Carolina, John earned his bachelor’s degree in chemistry at Johnson C. Smith University and then served in the U.S. Army at Fort Belvoir in Virginia for two years before coming to NIST in 1957.
He certainly wasted no time while in the D.C. metropolitan area. While in the Army and then at NIST, John took graduate-level courses at American University and Howard University and operated multiple businesses, including a gas station and a home improvement store. Although he was an entrepreneur outside of the lab, John still made quality time for family. He and his wife were a couple in every way, including driving to work together.
Many of the publications he authored related to an all-new measurement method called galvanostalametry. He and his colleague Abner Brenner came up with this unique approach to determine the concentrations of metal ions in liquids, which could be useful for measuring the amount of lead or other heavy metals in water.
To understand the concepts at play, look to this diagram from a paper by John and Abner. The U-shaped apparatus had a narrow tube on one side and larger pocket on the other. Liquid filled the narrow tube almost to the top, and it filled some of the larger pocket as well.

Does something look … off?
Take another look, specifically at the liquid. Under normal circumstances, due to the pull of gravity, you would expect it to pool at the bottom of the device.
But the circumstances weren’t normal. The device was under vacuum, sealed up with only water in both its liquid and vapor form. Inside the narrow tube, the forces between the water molecules and glass surface supported the weight of the water column, balanced against the force of gravity.
The arrangement was only barely stable. Creating just one tiny gas bubble in the tube could break the bonds between liquid molecules and the glass, unleashing the whole column of water and causing it to splash down. The formation of this gas bubble is known as “cavitation,” because the bubble’s spherical shape contains a hollow space, or a cavity.
John and Abner built on early work in cavitation by former NIST director Lyman Briggs, who passed an electrical current through the system to separate the water molecules into their gaseous, column-collapsing components. But their new idea wasn’t solely to break the water molecules apart and trigger the collapse.
To John and Abner, timing was everything. They realized that, when running the experiment using a liquid that contained more than just water, the electricity would interact with the other particles in the liquid first. Knowing how long it took for the current to flow through the other material (before it ripped the water molecules apart and triggered the column collapse), the research duo could determine the amount of the other substance in that water sample.
Ultimately, galvanostalametry didn’t make its way into our chemistry textbooks for a few reasons. Although it could compete with other techniques studying the same thing at the time, it was still tedious to repeat the process of creating the water column for each experiment. Also, the advent of more advanced electronics surpassed this creative electrolysis method.
A Personal Note, and a Plea
The fate of John and Connie hit close to home precisely because of how little known it was to me. My mother grew up in Wheaton and feels a deep connection to the community. Her side of the family makes a point at gatherings to discuss what they remember of the area in the 1970s and 1980s.
But nobody in my family, nor any of their friends from Wheaton, could recall ever hearing of the incident that took John and Connie. Only in the last few years, when information about it was posted to a local Facebook group, did we learn of it.
And both men worked at NIST, where I’ve now worked for almost three years and where my mother also once worked.
So, relying heavily on the help of John’s and Connie’s families, along with our own scientists, research librarians and alumni association, I began to dig. This blog post is the result, but there are always more questions. What brought Connie and John into science and chemistry? What hopes did they have for the future of their fields? Where could their efforts have taken them?
… And maybe that’s where you come in.
A Facebook post was enough to set me looking for context, so perhaps a blog post can do the same. If you knew John or Connie, please comment below and share your memories of them.
About the author
Related Posts
Comments
Thank you, Riley, for calling this to our collective attention. It is indeed a sad, remarkable truth that this story of two NIST scientists, gunned down in a hate crime committed within many of our lifetimes is largely unknown to us. This event was a human tragedy, an outrageous act of violence, and relevant in so many ways to our ongoing awakening to the many systematic biases that exist in what is deemed memorable that I hope we all will remember these names and ask ourselves how it came to be that we collectively did not know them as an important part of our history.
It is heartbreaking that this country and the families of these two talented scientists were deprived of their love and skill because of racism and white privilege. Fifty years later, I wish everyone would read or be taught from the book "The 1619 Project", rather than allowing legislatures to make it harder for black citizens to vote.
I never heard of this. I was an NSF intern in a program run by Leo Schubert at AU for high school seniors. Was assigned to liquid chromatography branch doing work on polycyclic arenes with a highly decorated staff of chemist. I wasnt that interested and hung out at a concession stand down the hall run by a blind cashier, who was liste ing to the Watergate hearings. In our office was a Black chemist named Willie who helped me along. Been a long time.
Thanks for the note! Is our former director the Willie that you're referencing? https://www.nist.gov/people/willie-may
I was a grad student working in the analytical spectroscopy area at that time. I recall the name Connie Stanley but not much else from that period. I sort of remember the event but it was too long ago to recall details. Since our work involved SRMs I likely met him. If he was in the NBS bowling league, I might also recall him from there. That chromatographer was indeed Willie May. The concession stand was at the end of the first floor corridor in building 222. The organic group was on that floor.
Very interesting and horrifying story.
Arriving at NBS in 1982, I did not know John Sligh, Connie Stanley, nor had heard of the crime, but had heard of galvanostalametry and of Abner Brenner, whom I met in his later years in connection with other work on electroless deposition of nickel-phosphorous (NiP) alloys. I do not know if John Sligh was involved in the NiP work, which later found significant applications in manufacture of hard disk drives (see Wikipedia, https://en.wikipedia.org/wiki/Electroless_nickel-phosphorus_plating).
Sligh (first author) and Brenner's second article on galvanostalametry, is at J. Electrochem. Soc. vol. 114(5), pp 461-465 (May 1967 issue). Also see pp 5-8 of Brenner's 1962 Blum lecture at https://www.nmfrc.org/pdf/2018/04abner_brenner1962.pdf for reference to Sligh's initial work. A Google search of "galvanostalametry" yields downloadable files for both.
The Blum lecture indicates that Sligh discovered the phenomenon. Brenner was presumably his manager (thus got to present the 1962 Blum lecture, which also covered other areas). Per that lecture, Sligh had personally previously reported on the discovery of galvanostalametry to the Electrochemistry Society at its May 1962 meeting. A search of J. Electrochem. Soc. at the NIST Library should give the relevant references, including the lecture program of the May 1962 meeting.
In galvanostalametry, at the upper (indicator) electrode there is a net negative pressure (corresponding to the height of the liquid column). The column is held up only by surface tension. Any microbubble that forms at the top causes the column to collapse suddenly. Such a bubble is forms by electrolysis of the water, thus forming H2. If an electroactive species is dissolved in the water, it will be reduced first, preferentially to the water. So the H2 bubble does not form immediately. The initial electrolysis of the water only occurs after the electroactive species is exhausted in the region immediately surrounding the upper electrode. Only then does the H2 bubble form, and the column collapses. Thus, the time to the collapse is a function of the concentration of the electroactive species in the solution filling the cell.
As noted by Sligh and Brenner in the 1967 J. Electrochem. Soc. article, the process is analogous to chronopotentiometry. In chronopotentiometry, a step in potential occurs at the point of depletion of the dissolved electrolyte. This potential step is measured as a function of time.
In contrast, in galvanostalametry, the time of collapse is determined. The galvanostalametric process is difficult to set up (I imagine it as somewhat analogous to avoiding foaming of a carbonated beverage on opening of a pop can). So chronopotentiometry is easier and more exact, and it is still used.
Kenneth Pratt (NBS/NIST retired, 2016)
Thannks for telling
My wife and I attended a social event with Connie Stanley the weekend prior to his untimely death.
Connie was my Uncle. He was the brother of my Grandfather. Everyone in the car with Connie are related to me.






Unfortunately I have no additional information to add, but I want to thank the author and the families for sharing the stories of these two scientists.