Taking Measure
Just a Standard Blog

Biopharmaceutical manufacturing uses living cells to produce therapies that treat diseases like cancer, diabetes and autoimmune disorders. Manufacturing medicine using biology presents different challenges from the traditional chemical manufacturing processes that stamp out identical pressed pills. Biomanufacturing processes are hard to control, and the products are difficult to define as “identical” from batch to batch.
Despite these challenges, biopharmaceuticals are critical to public health because the advantages are significantly greater. Scientific understanding of diseases and the success of biologically manufactured therapies to treat them has increased dramatically. But it can take a decade from design to full production of a biopharmaceutical — not fast enough to meet the needs of all the patients, or to beat competition from overseas.
The National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) was funded by the National Institute of Standards and Technology (NIST) in 2016 to address those challenges through innovation. The stakes are high. This industry is critical for the health of our economy: The biopharmaceutical industry represents more than 290,000 jobs, provides biopharma manufacturing workers with an average annual wage of $115,010, and contributes $1.3 trillion to the U.S. economy.
Helping the U.S. biopharmaceutical industry innovate and lead the world is my (day, night and weekend) job as director of NIIMBL, one of the 14 public-private manufacturing innovation institutes that make up Manufacturing USA®. I’m also the current chair for the Manufacturing USA Institute Directors’ Council, which adds another dimension to my reflections on how these U.S. innovation centers can deliver maximum impact.
Approaches to the Biopharma ‘Missing Middle’
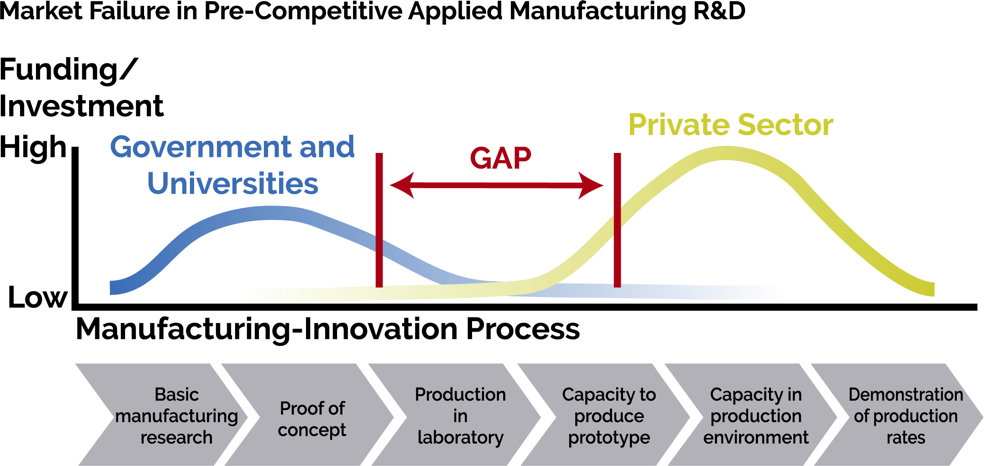
Which brings me to the gap. The metaphorical gap is the manufacturing “valley of death” between a good idea and a successful product. The U.S. leads the world in innovations and inventions, yet the manufacturing capabilities and new products are often “stranded in the lab,” or get developed in other countries. NIIMBL and the other manufacturing institutes focus on ensuring that American innovations get made in America.
The literal gap is the one that I minded between the train and the platform during a week of travel around the U.K. with a few colleagues from NIST and Food and Drug Administration (FDA) to learn how our counterparts advance biomanufacturing innovation across the pond. Most of the big pharmaceutical companies have manufacturing and R&D sites inside the U.S. and U.K., and both our governments are concerned about the potential for companies to relocate those operations and the jobs that go with them to other countries.
The biggest argument for keeping operations in the U.S. is to ensure consistent product quality, as biomanufacturing processes are finicky: Everything from the water supply to the skills of the technicians matters. The global industry is faced with choices on investing for the future, and both countries want to tilt in favor of domestic manufacturing, including “upskilling” the workforce and ensuring solid domestic supply chains. To borrow a phrase from FIRST Robotics, the relationship between NIIMBL and its U.K. counterparts is really one of “Coopertition,” where there is opportunity in working together to make biomanufacturing more cost-efficient and nimble while keeping each of our domestic missions in mind.
Over five days in five cities, we met with colleagues with the Cell and Gene Therapy Catapult, the Centre for Process Innovation (CPI) National Biologics Manufacturing Centre, the Industrial Biotechnology Innovation Centre and the Medicines Manufacturing Innovation Centre, as well as the BioProNET and the new Vaccines Manufacturing and Innovation Centre, all of which are part of the U.K.’s Life Sciences Industrial Strategy to “Build a Britain Fit for the Future.”
Through our discussions, we learned how each of these centers is taking a different approach to bridging that gap between R&D innovation and manufacturing to strengthen the U.K.’s position in the global bioeconomy. While I’m still processing much of what we learned, here are a few themes from the U.K.’s approach to innovation for reflection.
Each center defines its mission space as the gap between what is needed and the capabilities offered by others
As one of the center directors stated: “If we find ourselves competing with someone else who can do it, then we are in the wrong space.” These centers are positioned to move new processes to a maturity level where a commercial organization will accept the remaining risk for the last stages of development and get the product into the market.
Bridging the gap requires more than technology innovation
CPI, an independent technology innovation center in the U.K., leads small companies through an “Innovation Integrator” program (somewhat like NIST’s Baldrige Performance Excellence Program) and helps start-up biotechnology companies understand all the requirements for success, from science to personnel to intellectual property. These innovation centers help entrepreneurs understand not only how to design a pilot-scale manufacturing process, but also how to calculate scale-up costs, which often dictate the path forward for a small company.
Access to infrastructure is also critical. The growth of cell and gene therapy manufacturing keeps outpacing the capacity, and that must be addressed to reduce drivers for sending manufacturing offshore. On the day of our visit, the Cell and Gene Therapy Catapult announced that it had doubled its pilot-scale manufacturing capacity. That space is critical for small and medium companies, which are less resilient than large companies yet carry much of the risk in bringing novel products through the valley of death in highly regulated applications.
Access to capital is a major hurdle for small companies as well. The U.K. centers actively promote network connectivity between investors and entrepreneurs and help their members identify potential sources of funding appropriate for different stages of maturation.
Another key gap for advanced manufacturing sectors is the availability of a skilled workforce. Our U.K. colleagues understand, as we do in Manufacturing USA, that there are more unfilled biomanufacturing jobs than there are skilled workers to fill them. On both sides of the Atlantic, we are partnering with universities, community colleges and economic development organizations to both fill the pipeline for the future and provide further training for the existing workforce.
When innovation matters most
The U.K. centers also work with industry to determine whether the existing state of the art for any given process needs disruption, automation or perhaps just better duct tape. Pulling out the shared innovation needs can be challenging in an industry that tends to regard all knowledge as a competitive asset. NIIMBL and our U.K. counterparts must constantly build trust so we can bring partners to the table to develop robust and scalable technical solutions that advance the industry as a whole.
Don’t forget the practitioners and regulators
Our U.K. colleagues know that bringing novel therapies out of clinical labs and small biotech companies to the patients who need them requires a full ecosystem approach that includes the clinical practitioners and health regulators. The manufacturing innovation centers partner with clinical innovation centers and the U.K.’s Medicines and Healthcare Regulatory Agency in myriad ways.
On our side of the pond, the U.S. FDA also recognizes the benefit of working with stakeholders in the biopharmaceutical manufacturing ecosystem to accelerate innovation. The FDA has engaged with NIIMBL in multiple ways, including having Cmdr. James Coburn as a liaison between NIIMBL and the Office of the Chief Scientist at FDA, as well as an interagency “loan” to NIST of Jeffrey Baker. Jeff is helping map out ways for regulatory scientists to work collaboratively with NIIMBL to accelerate acceptance of new technology into biomanufacturing processes without creating conflicts of interest in their roles as regulators.
Leverage all relationships to tackle the big problems
The U.K. innovation centers have different operating models, technology scopes and professional cultures, but benefit from understanding the role each plays in alignment with the U.K.’s Life Sciences Strategy. The centers collaborate, learn from each other, and are rewarded in funding allocations for collaborating. Our Manufacturing USA program is developing those connections, and my trip to the U.K. has increased my commitment to invest the time to strengthen NIIMBL’s relationship with the other institutes for mutual benefit.
NIIMBL also enjoys collaborating with our colleagues at NIST, who appear to have industry responsiveness encoded in their DNA. NIST and NIIMBL are natural partners, both focused on pre-competitive “wins” that lift all boats — and NIIMBL industry partners know the value of NIST measurement science in understanding their complex and highly regulated products. It is also evident that there is genuine goodwill between my NIST and FDA colleagues to work together to help each agency accomplish its respective mission. The U.S. government is a big enterprise, and my week in the U.K. reminded me how complex institutional silos can be navigated by people trying to do the right thing for the good of the nation.
After my week minding the gap, I have a greater sense of partnership with my U.K. colleagues and look forward to continuing discussions about how we might work together. We all focus on generating solutions for broad industry challenges, developing new skills, building an innovation ecosystem, and increasing manufacturing capacity and innovation. Like NIIMBL, the centers are designed to be at the cutting edge doing difficult things that no one can do, or do alone, because of risk.
One question we asked each U.K. center was how it assesses its effectiveness: Their most valuable indicator is continued willingness of industry partners to collaborate with them. That same indicator is my gut check leading our NIIMBL team as it works to deliver that value for our 120-plus members.
I am more energized than ever for the work ahead. In the coming months, we’ll be moving into our new official headquarters at the University of Delaware. This state-of-the-art facility will drive new opportunities for innovation within the biopharma community, through access to equipment for process innovation, technology platform demonstrations, and NIIMBL-led collaborative research projects. If you want to join us in this work, please leave a comment!
About the author
Related Posts
Comments
I was diagnosed with the mysterious disease Fibromyalgia in 1992. It is 2020 and I am being treated with the same medication as I was then. I am no better today than I was then. I have finally found a Clinical Trial that is hopeful for me and I am waiting to get started. I like what I just read about the challenges of you and your team, and I would like to throw you a patient’s concern when it comes to participation in a trial. Being disabled and living on SSDI, I am finding it a challenge for me to find a way to transport myself to the trial site. Please don’t overlook the needs of willing participants. I am willing to give of myself to help millions of others, but getting there might be something I cannot do. Is medical science willing to miss out on people like me or can we come together and make this happen? Can we make this work for everyone? Just a thought and something I think is important for everyone.
I think all of us in biopharma are working to figure out the best way to close this gap. Great thoughts on a lot of the topics we are working on at my company. Love to learn more on what NIIMBL is working on.
Jeremiah Genest
Head of Digital Quality
Viral Vector Services
Thermo Fisher Scientific
[email protected]
Hi Dr. Lee, This was really interesting to read. NIIMBL and your vision can surely close this gap!
I want to thanks for your time for this wonderful Article!! I definitely enjoying every little bit of it and I have you bookmarked to check out new stuff you blog post.
If more people that write articles really concerned themselves with writing great content like you, more readers would be interested in their writings. Thank you for caring about your content.





I was not completely surprised by the focus on the economy and keeping people employed; I would like to see increased focus on the patients.