Summary
Starting in 2013 the Army Research Laboratory (ARL) along with the University of Maryland (UMD) College Park led the foundation of a battery research consortium that was later named the Center for Research on Extreme Batteries (CREB), with NIST as one of the initial partners. Currently CREB is actively working on research projects funded through a grant from ARL. CREB is currently focused on advancing battery technology for the U.S. Army. NIST’s Neutron and X-ray Tomography (NeXT) system exploits the complementarity of the interactions of neutrons and X-rays with matter to enable clear identification of the electrochemical processes that are active in the battery to help solve existing safety and lifetime problems with lithium-ion batteries.
Description
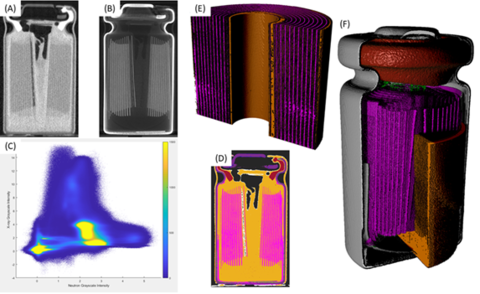
Figure 1: Commercially available 10180 lithium nickel manganese cobalt (LiNMC) battery (A) neutron reconstruction midplane slide, (B) X-ray reconstruction midplane slice, (C) bivariate histogram with neutron grayscale values along X-axis and X-ray grayscale values along Y-axis, (D) colorized midplane slice from segmentation based on bivariate histogram, (E) 3D image of electrodes from segmentation, and (F) 3D cutaway image of the battery showing metal case (gray), polymer gasket (red), copper current collector and NMC electrode (magenta), and graphite electrode and electrolyte (orange) [2].
The NIST Neutron Imaging Facility (NNIF) has developed an in situ simultaneous Neutron and X-ray Tomography (NeXT) capability that allows for 3D imaging of batteries and other objects [1-3]. Neutrons are complementary to X-rays and when combined allow one to segment and identify features to non-destructively track the time evolution of a battery to the point of failure under the extreme environmental conditions that soldiers will find in the field as we see in Figure 1. Due to the large absorption cross-section of the isotope lithium-6, neutrons can detect concentrations and plating of lithium. Specially designed environmental chambers can simulate real world field conditions as both neutrons and X-rays can penetrate through the chamber windows to image a battery being cycled and subjected to extreme conditions. In addition to lithium, neutrons are also able to image hydrogenous materials in the presence of metals allowing study of alkaline batteries.
Neutron imaging can follow lithium plating during charge/discharge cycling [4-7]. Alternatively, batteries built with pure lithium-7 allow neutrons to see changes in the electrolyte during fast discharge. NIST will continue to work collaboratively on various battery imaging projects with CREB partners. As needed we can create detection standards for quantification of lithium to guide the analysis of data.
Associated Battery Testing Equipment
- Arbin Mixed LBT Battery Cycler
- 8 channels High current
- 0-5 V
- Current Ranges:
- 1st current range: -60 A to +60 A
- 2nd current range: -5 A to +5 A
- 3rd current range: -500 mA to +500 mA
- 4th current range: -20 mA to +20 mA
- 300 W maximum continuous power per channel
- 8 channels low current
- 0-5 V
- Current Ranges:
- 1st current range: -1 A to + 1 A
- 2nd current range: -50 mA to +50 mA
- 3rd current range: -2 mA to +2mA
- 4th current range: -100 µA to +100 µA
- 5 W maximum continuous power per channel
- 16 T-type thermocouple inputs
- Integration with Gamry Reference 3000 for full EIS capabilities
- 8 channels High current
References
[1] J. M. LaManna, D. S. Hussey, E. Baltic, and D. L. Jacobson, “Neutron and X-ray Tomography (NeXT) system for simultaneous, dual modality tomography,” Rev. Sci. Instrum., vol. 88, no. 11, p. 113702, Nov. 2017, doi: 10.1063/1.4989642.
[2] J. M. LaManna, D. S. Hussey, V. H. DiStefano, E. Baltic, D. L. Jacobson. NIST NeXT: a system for truly simultaneous neutron and X-ray tomography. In: Hard X-Ray, Gamma-Ray, and Neutron Detector Physics XXII, edited by Fiederle M, Burger A, Payne SA. SPIE, p. 24., doi: 10.1117/12.2569666.
[3] G. V. Riley, D. S. Hussey, and D. Jacobson, “In Situ Neutron Imaging Of Alkaline and Lithium Batteries,” Apr. 2010, pp. 75–83, doi: 10.1149/1.3414005.
[4] J. B. Siegel, X. Lin, A. G. Stefanopoulou, D. S. Hussey, D. L. Jacobson, and D. Gorsich, “Neutron imaging of lithium concentration in LFP Pouch cell battery,” J. Electrochem. Soc., vol. 158, no. 5, 2011, doi: 10.1149/1.3566341.
[5] J. P. Owejan, J. J. Gagliardo, S. J. Harris, H. Wang, D. S. Hussey, and D. L. Jacobson, “Direct measurement of lithium transport in graphite electrodes using neutrons,” Electrochim. Acta, vol. 66, pp. 94–99, Apr. 2012, doi: 10.1016/j.electacta.2012.01.047.
[6] C. Cai, Z. Nie, J. P. Robinson, D. S. Hussey, J. M. LaManna, D. L. Jacobson, G. M. Koenig. "Thick Sintered Electrode Lithium-Ion Battery Discharge Simulations: Incorporating Lithiation-Dependent Electronic Conductivity and Lithiation Gradient Due to Charge Cycle," J Electrochem Soc 167: 140542, 2020, doi: 10.1149/1945-7111/abc747.
[7] Z. Nie, S. Ong, D. S. Hussey, J. M. Lamanna, D. L. Jacobson, G. M. Koenig. Probing transport limitations in thick sintered battery electrodes with neutron imaging. Mol Syst Des Eng 5: 245–256, 2020, doi: 10.1039/c9me00084d.

