Small variations in bottlebrush polymers result in measurable changes
NIST team scores firsts in illustrating changes and isotopic labeling
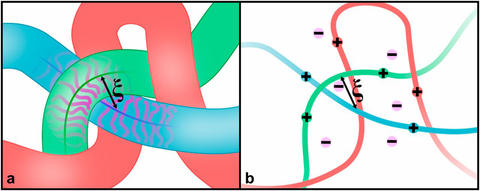
Illustration of the mesh formed by bottlebrush polymers (a) and polyelectrolyte solutions (b). The NIST team found that their properties are similar despite differences in their chemistry.
NIST scientists have found that very small changes in the structure of bottlebrush polymers prepared by “precision synthesis” result in measurable differences in the structure and behaviors of these materials. The NIST researchers combined synthesis, experimentation, and modelling to investigate bottlebrush polymers, the name of which evokes their structure of sidechains packed densely along a backbone. Their results were published in the February 24, 2020 issue of the Proceedings of the National Academy of the Sciences.
Bottlebrush polymers are a new class of materials with applications in specialty areas that demand advanced function and performance, like photonics. Ultrasoft bottlebrush polymers are also suited to mimicking biological tissues and can exhibit superior vibration dampening, among other uses.
The NIST team also found that precision synthesized bottlebrush polymers have the same packing, and ability to scale their packing in response to structure changes, as solutions of more common and much-studied polyelectrolytes, charged polymers with no side chains at all.
Because of their dense structure, the molecules of bottlebrush polymers have been difficult to fully measure. The NIST team used precision synthesis, a production method that results in “very explicit architecture with all the molecules close to the same length,” as co-author Kathryn Beers describes them. Comparatively, the polymers we use every day that are produced on a large scale, are “a hot mess” of lengths and structures says Beers, but nevertheless fit for their purposes as food packaging or textiles.
In a first for the precision synthesis of bottlebrush polymers, the NIST team used isotopic labeling: They selectively swapped out hydrogen molecules for deuterium, an isotope of hydrogen that scatters energy differently, helping the researchers more confidently determine molecular packing. The energy was supplied by the NIST Center for Neutron Research’s small angle scattering instrument. The team analyzed the structures of three families of synthesized bottlebrushes with variations in their density, from one sidechain every two backbone carbons, to one sidechain every five backbone carbons, to two sidechains every five backbone carbons. This work shows for the first time that these very small changes in structure—just every few carbon atoms—changes the characteristics of bottlebrush polymers. In this case, the more space between sidechains in the synthesized bottlebrushes, the less regularly they pack.
These results were echoed by computer modelling also performed at NIST. Even more interesting is that those densely packed side chains force packing and order of the molecules in the same way that charge and salt concentration force charged polyelectrolyte chains to pack. A direct comparison was made between the increase in density and length of those side chains and the repulsion of charge. This work was only possible because the NIST team’s synthesis methods were able to control very small changes in the bottlebrush polymer chains.
Compared to charged polymers, bottlebrush polymers are easier to process and characterize. Showing that bottlebrush polymers have the same desirable properties as charged polymers may help others realize new applications for these materials.
Paper: Joel M. Sarapas, Tyler B. Martin, Alexandros Chremos, Jack F. Douglas, and Kathryn L. Beers. Bottlebrush polymers in the melt and polyelectrolytes in solution share common structural features. Proceedings of the National Academy of Sciences, Published February 24, 2020. DOI: https://doi.org/10.1073/pnas.1916362117

