
Alexandra Rodriguez demonstrating the broken contact style of data logger.
Recipients of an annual flu shot may be surprised to learn that there is currently no official standard for vaccine storage equipment in clinics, pharmacies, and other health providers' offices – a potential problem, since vaccines need to be kept within strict temperature limits to remain viable.
What's at stake is money as well as health, says Michal Chojnacky of NIST's Physical Measurement Laboratory (PML), Sensor Science Division. "Vaccines can go bad if stored improperly. If this happens and no one detects it, you run the risk of delivering ineffective vaccines to patients," she explains. "The other danger is thinking that a vaccine goes bad when it doesn't. And if that happens then you waste the cost,* and you potentially delay vaccines getting to the intended patients."
Fortunately, a standard is on the way. Chojnacky is one of about 30 members of a multi-institution committee whose goal is to define a set of performance standards for vaccine refrigerators, freezers, and other storage devices, to help providers choose the best equipment for the job. The committee, formed in 2015 and run by NSF International (formerly the National Sanitation Foundation), includes representatives from the Centers for Disease Control and Prevention (CDC), state health department immunization programs, non-profit organizations, and vaccine storage equipment manufacturers.
"It is not always clear whether a particular refrigerator or freezer will meet a health care provider's needs," Chojnacky says. "The creation of an NSF standard for vaccine storage equipment will serve to demystify this selection process for the nurses, pharmacists, and other health workers who administer vaccines."
To create the new standard, the committee needs to know how providers use their vaccine storage units. But as it turns out, there are no reliable answers to even basic questions about daily usage.
To give the committee some data to work with, Chojnacky and Alexandra Rodriguez, a third-year chemical engineering undergraduate student at Drexel University in Philadelphia, Penn., have been conducting an exploratory study with a sampling of clinics around the country. They are asking a simple question: how often do vaccine providers open their refrigerator doors?
Chojnacky and Rodriguez are completing round one of their study, which includes about 35 vaccine storage refrigerators from two dozen pediatric clinics around the country. The clinics were given a data logger per fridge. Each logger records when the refrigerator door is open using either a light sensor inside the fridge or, for glass-front units, a device that records when contact is broken between fridge and door. (The loggers do not document temperature changes.) Participants used the loggers for about 2 weeks each and filled out a questionnaire, which included information about the site and how many vaccines were distributed each day.
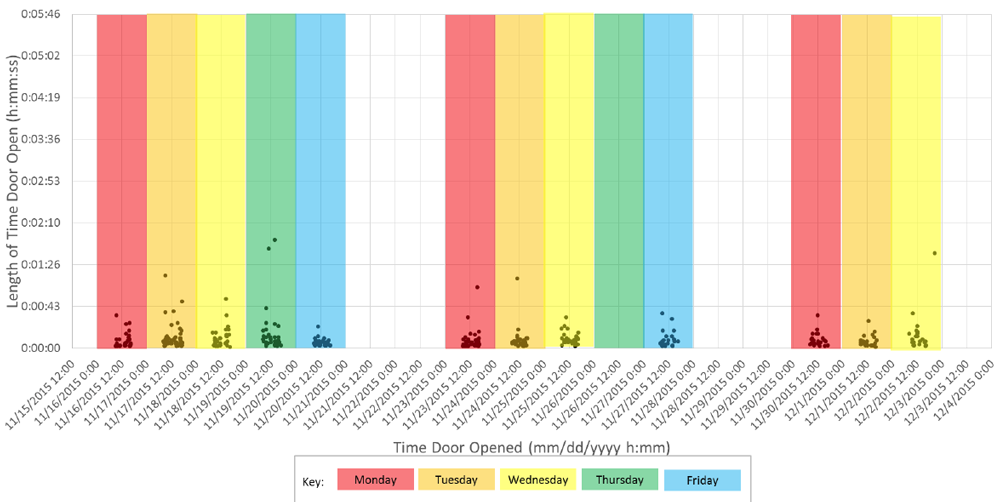
Though their sample size is small, the information is already helping Rodriguez and Chojnacky refine their ideas for future studies. For example, the results so far show that in daily use, fridges are open for an average of 7 seconds at a time, which is less than they'd expected. At the busiest of clinics, units were accessed as many as 33 times in one hour, though the average time between door openings was about 3 and a half minutes.
Previously, the PML team had done some general temperature tests in which fridge doors were open for 30 seconds every five minutes. Using this new information, the PML researchers can repeat their tests in their own lab with what may be a more realistic open-door duration and frequency.
"The strange and unique thing we found working on this was that there aren't – as far as we know – a lot of people doing testing like this," Chojnacky says. Much more often, research is focused on pre-distribution vaccine environments, or strategies for keeping vaccines cold when no refrigeration is available.
The NSF committee's ultimate goal is to create a protocol that will be used to test refrigerators and freezers to make sure they are appropriate for vaccine storage. Storage units that pass the tests would be granted an NSF certified seal.
In March, the PML team will meet with the rest of the NSF committee to discuss their results and make plans for round two of the fridge door study, which will include vaccine storage units in commercial pharmacies and other types of outpatient clinics.
-- Reported and written by Jennifer Lauren Lee
*For examples of the going rate for vaccines, see the CDC's vaccine price list.

