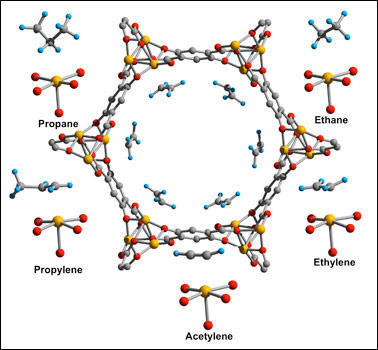
Iron projecting into the pore of the tubelike metal-organic framework (center, looking down its roughly nanometer-wide opening) attracts the light hydrocarbon molecules that surround it to varying degrees. These varied attraction levels could make the framework more efficient at hydrocarbon separation than current refinery processes.
Measurements taken by a team including National Institute of Standards and Technology (NIST) scientists show that a newly devised material has the ability to separate closely related components of natural gas from one another, a task that currently demands a good deal of energy to accomplish. The results, published March 30, 2012, in the journal Science, might improve the efficiency of the distillation process.
The material is a new type of metal-organic framework (MOF), a class of materials whose high surface area and tunable properties make them promising for applications as varied as gas storage, catalysis and drug delivery. This particular iron-based MOF, which the research team refers to as Fe-MOF-74, was built in the lab of Jeffrey Long, a professor of chemistry at the University of California Berkeley, and analyzed by the team at NIST and the Australian Nuclear Science and Technology Organisation's Bragg Institute.
Natural gas taken straight from the ground consists of a complex mixture of molecules called hydrocarbons, only some of which are needed for use in any specific product such as fuel or plastic. Separating the lighter types of hydrocarbon from one another—propane and ethylene, for example—is difficult because their weights are so similar. Currently, the most effective separation method involves chilling light hydrocarbons down to the point where they all liquefy, sometimes as low as 100 degrees below zero Celsius, and waiting until the heavier liquids settle below the lighter ones.
"A good percentage of the energy needed for separation goes to the cooling process," says Wendy Queen, a postdoctoral fellow at the NIST Center for Neutron Research. "A material that can selectively adsorb light hydrocarbons could offer significant energy savings, making separation more economical."
Through a microscope, Fe-MOF-74 looks like a collection of narrow tubes packed together like drinking straws in a box. Each tube is made of organic materials and six long strips of iron, which run lengthwise along the tube. The team's analysis shows that different light hydrocarbons have varied levels of attraction to the tubes' iron, a finding that can be exploited for separation. By passing a mixed-hydrocarbon gas through a series of filters made of the tubes, the hydrocarbon with the strongest affinity can be removed in the first filter layer, the next strongest in the second layer, and so forth.
"It works well at 45 degrees Celsius, which is closer to the temperature of hydrocarbons at some points in the distillation process," Queen says. "The upshot is that if we can bring the MOF to market as a filtration device, the energy-intensive cooling step potentially can be eliminated. We are now trying out metals other than iron in the MOF in case we can find one that works even better."
E.D. Bloch, W.L. Queen, R.Krishna, J.M. Zadrozny, C.M. Brown and J.R. Long. Hydrocarbon separations in a metal-organic framework with open Iron(II) coordination sites. Science, March 30, 2012. DOI:\10.1126\science.1217544

