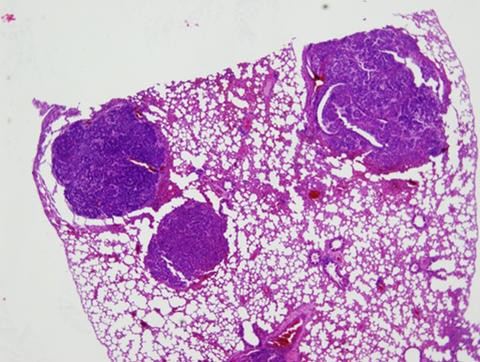
Section of lung tissue from a double knockout mouse, stained to show lung tumors. Mice lacking both the neil1 and nth1 genes were particularly prone to pulmonary anbd liver tumors during their second year.
Deleting two genes in mice responsible for repairing DNA strands damaged by oxidation leads to several types of tumors, providing additional evidence that such stress contributes to the development of cancer. That's the conclusion of a recent study in DNA Repair by researchers at the National Institute of Standards and Technology (NIST), Oregon Health and Science University (OHSU) and the New York University School of Medicine (NYUSM).
Although all cells need oxygen to survive, the element also can be stressful to cells and their components—particularly DNA—as part of "reactive species" in the environment, such as free radicals and peroxides. The damage levied on DNA by these compounds can include lesions, breaks, cross-links and deletions—errors in our normal genetic codes that, if left unchecked, may accelerate the aging process and increase susceptibility to several disease states. In humans, DNA repair genes produce enzymes called DNA glycosylases that excise sections of DNA strands already modified by oxidative stress, and thus protect the genetic material.
One of these repair genes, neil1, was identified and characterized in 2002 by Sankar Mitra and his team at the University of Texas Medical Branch in collaboration with NIST researchers Miral Dizdaroglu and Pawel Jaruga. The gene produces a DNA repair protein, NEIL1 that is nearly identical in humans and mice. Therefore, a mouse serves a perfect model for studying the biological function of the neil1 gene in both species.
To do this, OHSU researchers under R. Stephen Lloyd genetically engineered mice without the neil1 gene (known as neil1 knockout mice). During their first 6-10 months of life, the majority of male mice developed severe obesity, dyslipidemia (abnormal levels of lipids in the blood), fatty liver disease and hyperinsulinemia (excess levels of circulating insulin in the blood). In humans, these disorders are collectively known as metabolic syndrome, a condition that affects more than 40 million persons in the United States.
In collaboration with Dr. Lloyd's group, a second research team under George W. Teebor at NYUSM engineered mice that were missing either the neil1 or the nth1 gene (nth1 encodes for another DNA glycosylase, the NTH1 protein) or both these genes. These latter are known as neil1/nth1 double knockouts. NIST's Dizdaroglu and guest researchers Pawel Jaruga and Güldal Kirkali found that both types of knockout mice exhibited significant accumulation of two lesions called formamidopyrimidines in the DNA of the liver, kidney and brain. This indicates that there was a lack of DNA repair in these organs.
During the second year of life, both types of mice also developed pulmonary and hepatocellular (liver cell) tumors. Double knockout mice had a higher incidence of tumors than the single knockouts.
The researchers state that their results emphasize the role of DNA repair in preventing carcinogenesis. The work may lead to the development of new measurement methods and reference materials for accurate and reproducible assessments of DNA damage and repair and contribute to understanding the role of oxidatively induced DNA damage and its repair in carcinogenesis. Future studies will focus on the role of NEIL1 in disease processes.
* M.K. Chan, M.T. Ocampo-Hafalla, V.Vartanian, P. Jaruga, G. Kirkali, K.L. Koenig, S. Brown, R. S. Lloyd, M. Dizdaroglu and G.W. Teebor. Targeted deletion of the genes encoding NTH1 and NEIL1 DNA N-glycosylases reveals the existence of novel carcinogenic oxidative damage to DNA. DNA Repair, Vol. 8, No. 7, pp. 786-794 (July 4, 2009).

