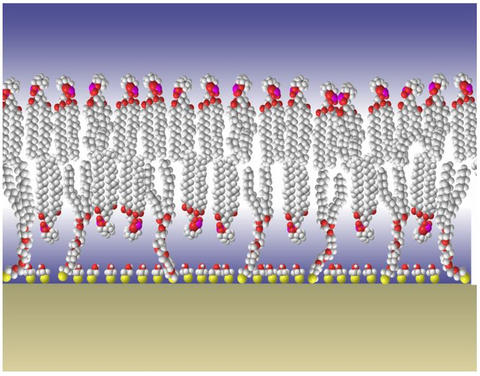
Diagram of NIST's tethered bilayer membrane model shows the silica surface covered with gold at the bottom. Sulfur atoms (yellow spheres) bind to the gold and act as anchors for the tethers, chains of atoms extending up to the lipid bilayer membrane at the top of the structure.
Researchers at the National Institute of Standards and Technology (NIST) and three collaborating institutions are using a new laboratory model of the membrane surrounding neurons in the brain to study how a protein long suspected of a role in early-stage Alzheimer's disease actually impairs a neuron's structure and function. The team's findings are reported in a new paper in the Biophysical Journal.
The brain's neurons transmit nerve impulses down a long stem that is surrounded by a two-layer membrane. In the neuron's normal, "rest" state, this membrane actively sorts sodium ions to the outside of the cell and potassium ions to the inside. To transmit a nerve impulse, an electrochemical change ripples down the membrane in advance of the impulse, making it temporarily more permeable and allowing the ions to swap places. That in turn changes the electrical potential across the membrane, allowing the impulse to pass. Afterwards, the membrane returns to rest and begins sorting the ions again.
Medical experts have hypothesized for years that small polypeptides called amyloid beta peptides somehow create a "leaky" membrane that disrupts this balanced back-and-forth switching of the electrical potential and, in turn, normal impulse transmission. Alzheimer's disease—the progressive brain disorder that is the nation's sixth leading cause of death—is believed to start with such breakdowns. As the disease progresses, amyloid beta peptides clump together to form plaques that further destroy nerve function.
Studying the beginnings of Alzheimer's is nearly impossible in humans because by the time the disease is diagnosed, most patients have moved into its later stages. Researchers at NIST have developed a laboratory model that recreates a simplified version of the nerve cell membrane, allowing the study of Alzheimer's disease mechanisms at the molecular level. A clever piece of molecular-level design, the system is built by first covering a silica surface with gold. Sulfur atoms, which bond well to gold, are then added to act as anchors to hold the bilayer membrane. The result is a stable, tethered membrane with an aqueous environment on both sides that accurately models the behavior of the nerve cell membrane.
A collaborative team of researchers from NIST, Carnegie Mellon University, the University of California-Irvine and the Biochemistry Institute (BCHI) in Vilnius, Lithuania, exposed the membrane model to different concentrations of a specific form of amyloid beta peptides comprised of soluble, tiny (5-6 nanometers, approximately twice the diameter of a DNA helix) chains. The researchers found increased cation movement across the normally strong barrier at the higher concentrations of the peptides. The data support the hypothesis that membrane "leakiness" is not due to a permanent hole being formed but rather to an aggregation of amyloid beta peptides in the membrane that allows cations to be passed from peptide to peptide across the bilayer, like a baton handed off by relay runners.
The researchers are continuing to use their model system to better understand the role amyloid beta peptides play in early-stage Alzheimer's disease. Future plans include investigating how amyloid beta peptide aggregates arrange themselves in the membrane, how the peptide aggregates affect or influence calcium channels (portals for calcium ion movement) in the membrane, and how the peptides interact with membranes constructed with other types of lipids.
G. Valincius, F. Heinrich, R. Budvytyte, D.J. Vanderah, D.J. McGillvray, Y. Sokolov, J.E. Hall and M. Losche. Soluble amyloid ß oligomers affect dielectric membrane properties by bilayer insertion and domain formation: Implications for cell toxicity. Biophysical Journal (published online June 13, 2008).

