Probabilistic Distribution of Dissolution Rate Constants for Cubic Tricalcium Aluminate in Water
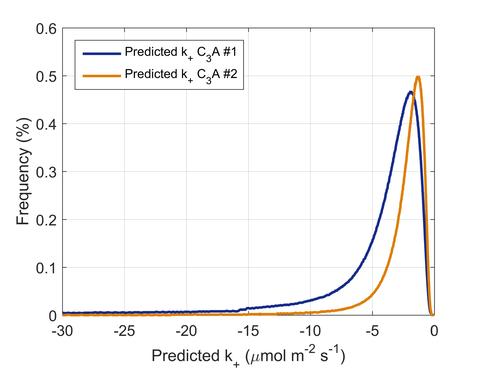
The probabilistic distribution of dissolution rate constants for cubic tricalcium aluminate (C3A-c) in water was quantified based on in-situ measurements of the changes in the nanoscale surface topography using reflection digital holographic microscopy. Measurements were performed on polished sintered pellets of C3A from two sources. C3A#1 consists of 94.2 % crystalline C3A-c, 2.0 % free CaO, and 3.8 % amorphous content. C3A#2 consists of 84.3 % crystalline C3A-c, 1.0 % free CaO, 4.6 % mayenite, and 10.1 % amorphous content.
DATA DOWNLOADS:
The data presented in the two text files, labeled C3A#1 and C3A#2, have two columns of values corresponding to the probabilistic distribution (histogram) data; the first column is the frequency and the second column is the rate constant value in units of μmol m-2 s-1.
All uncertainties associated with this dataset are discussed in an accompanying paper that is currently under review for publication.
Abstract for the accompanying paper:
In situ digital holographic microscopy is used to characterize the dissolution flux of polycrystalline cubic tricalcium aluminate (C3A-c) in flowing water. The results show that C3A-c dissolves at rates that vary considerably with time and location, implying a statistical distribution of fluxes. The dissolution flux from highly crystalline C3A-c depends exponentially on the water activity, with exponent of 5.2, and extrapolates to a median flux of −2.1 μmol m−2 s−1 with an interquartile range of 3.2 μmol m−2 s−1. The flux from a less crystalline source of C3A-c has a water activity exponent of 4.6 and a median flux of only −1.4 μmol m−2 s−1 with an interquartile range of 1.9 μmol m−2 s−1. These data suggest that the bulk dissolution rate of C3A-c can vary by at least 30 % from one source to another and that variability in the local rate within a single material is even greater because of the heterogeneous spatial distribution of structural characteristics (i.e., degree of crystallinity, chemical impurities, and defects).

