Summary
Protein drugs are the top selling pharmaceuticals worldwide, a hundred billion dollar industry, but also the fastest growing category of health care costs. The high cost of protein drugs can be traced to costly and inefficient manufacturing which lack the metrology tools necessary to improve the process and the product. US measurement science, standards, and reference data products provided by the NIST program in biomanufacturing will 1) benefit industry by providing measurement tools to aid in the characterization of their product, the manufacturing process, and product stability and 2) allow the FDA to develop scientifically sound regulatory guidance for development and manufacturing of safe and effective protein therapeutics. The impact of this program will be the development of lower cost biologic drugs and biosimilars, thereby allowing U.S. citizens affordable access to these innovative and life-saving medicines.
Description
The objective of the NIST biomanufacturing program is to support US biopharmaceutical industry delivery of high quality and low cost protein drugs around the world. The mission is fulfilled through the development of standards, measurement science, and state of the art tools that support advances in development, characterization and manufacturing of protein drugs and biosimilars (generic versions). The program was developed through close working relationships with members of the US biopharmaceutical industry, the FDA and international standards organizations (i.e. NIBSC, USP) to assess and authenticate current unmet and future measurement needs related to the manufacturing of protein drugs. To date NIST has sponsored seven workshops, involving these industrial and government stakeholders and established the "NIST Biopharmaceutical Measurement Roundtable" in 2012 to provide input and feedback on NIST programs. Three major areas of study (Protein Stability, Protein Structure, and Production Cell Variability) for metrology and standards development were identified and are the focus of the program.
Major Accomplishments
Biomanufacturing Related Standard Reference Materials (SRMs)
A key aspect of the NIST biomanufacturing program is provision of a variety of high quality reference materials and data products. Two initial SRMs are available or in development:
- IgG Standard Reference Material / Data (SRM/D)
NIST developed a recombinant IgG1κ monoclonal antibody (mAb) as a reference material for the biopharmaceutical industry, academia, regulatory authorities, and other standards organizations for a variety of uses such as: system suitability tests, establishing method or instrument performance and variability, comparing changing analytical test methods, and assisting in method validation. The candidate reference material was graciously donated by an industrial partner. The results from this exhaustive characterization were collated and published in a three volume ACS book series in 2014 and most of the data is publicly available online at http://igg.nist.gov. - Surrogate Protein Particle SRM
Protein particles, caused by aggregation of protein monomers, may limit drug stability and cause immunogenicity in patients, rendering the drug ineffective. Current methods used to assess these particles can vary by 10-fold in particle counts measured. A particulate SRM, used to normalize these disparate results, is being developed from a stable fluoropolymer material (ETFE) that mimics the optical properties, size, and shape of protein aggregates. The material has been evaluated for size and particle counts by 24 industrial and academic laboratories in a round robin study and will be certified by NIST for the number of particles per vial. A preliminary data analysis is complete and a draft report is in progress.
The NIST Biomanufacturing Program encompasses a suite of fundamental measurement science, standards, and reference data which enables more accurate and confident characterization of key attributes of protein drugs that are directly linked to their safety and efficacy. The initiative has three program goals related to characterization of protein therapeutics and their manufacture: Protein Stability, Protein Structure, and Production Cell Variability
Protein Stability
Protein stability is a property of therapeutic proteins that cannot be sufficiently measured at this time, but is critical for understanding the behavior of protein drugs and ensuring their safety and efficacy. Current projects cover the areas of protein particle sizing and counting (see SRM below), development of measurement methods and theory to predict protein stability in freeze-dried formulations and small volume rheology measurements.
Project links: |
|
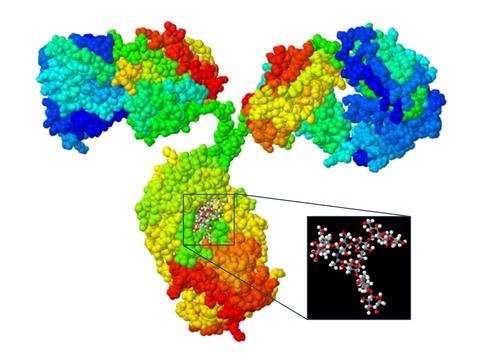
Protein Structure
Protein structure is another critical property that influence protein drug safety and efficacy. Current physico/chemical projects areas include: amino acid sequencing, post-translational modification characterization (e.g. glycan analysis), mass spectral analysis, NMR analysis and hydrogen/deuterium exchange MS characterization. In addition, an IgG SRM (see below) has been developed.
Project links:
|
|
Production Cell Metrology
Development of new measurement methods and tools for quantifying and understanding living cell populations will enable new strategies for engineering and controlling production cells to ensure consistent and effective manufacturing of protein therapeutics. Current projects include: authentication of cell lines using short tandem repeat (STR) DNA sequencing and cell line expression efficiency predictions based on fluorescence imaging measurements.
Project links:
|
|

