Matrix isolation infrared spectroscopy
Studies in the gas phase offer the potential for the most precise, detailed infrared spectroscopic measurements. However because of the high chemical reactivity of transient molecules, it is difficult to obtain gas-phase infrared spectra of them. Nevertheless, the well known advantages of Fourier transform infrared measurements, coupled with sophisticated digital data handling procedures, have permitted the acquisition of gas-phase survey spectra for a number of transient molecules. Diode lasers and other laser-based techniques with limited tunability have been used to obtain high resolution spectra of individual vibrational transitions of these species.
The application of matrix isolation sampling for the stabilization and spectroscopic study of reaction intermediates has recently been reviewed (see below). Because nitrogen and the rare gases are transparent through the entire infrared spectral region, matrix isolation measurements provide a potentially valuable survey tool. In these matrices, infrared absorptions are typically sharp, with half band widths between 0.1 cm-1 and 1 cm-1. Rotational structure is, with few exceptions, quenched. Multiple trapping sites occur, often resulting in the appearance of several absoption maxima — usually one or two of which predominate — over a range of a few cm-1.
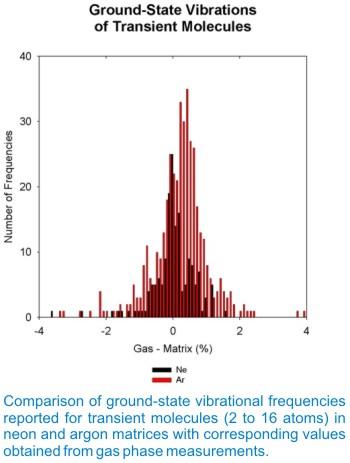
Matrix shifts for covalently bonded molecules trapped in solid neon or argon often are quite small. A comparison of positions of the ground-state vibrational fundamentals of over two hundred diatomic molecules observed in the gas phase and in nitrogen and rare-gas matrices has shown that, typically, the smallest matrix shift occurs for neon matrix observations, with successively greater matrix shifts for the heavier rare gases and for nitrogen. Except for very weakly bonded molecules and for the alkali metal and Group IIIa halides, matrix shifts of most diatomic molecules isolated in solid argon are smaller than 2%. Similar conclusions resulted from a comparison of neon- and argon-matrix shifts for the ground-state vibrational fundamentals of larger molecules. The generalization that matrix interactions are minimal for neon and that they increase as the mass of the rare gas is increased and become even more important for nitrogen and most other small molecule matrices is supported by experimental observations on larger molecules, as well. The figure on the right compares the observed matrix shifts for the ground-state fundamental vibrations of transient molecules trapped in solid neon and argon. For neon matrices, the maximum in the distribution lies near 0.0%, and for argon matrices, near 0.2%. For both neon and argon matrices, fewer than one-tenth of the matrix shifts are greater than 1%.
For more information, see the following review: The spectroscopy of molecular reaction intermediates trapped in the solid rare gases.
Related Links
Contacts
-
SSDWEB(301) 975-3727100 Bureau Drive
M/S 8440
Gaithersburg, MD 20899-8440

