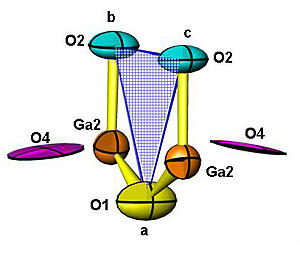
Researchers determined that a new material for fuel cells releases oxygen ions easily at low temperatures because many of the oxygen ions—marked here as O4—are not closely bound to the material's crystal framework.
Seeking to understand a new fuel cell material, a research team working at the National Institute of Standards and Technology (NIST), in collaboration with the University of Liverpool, has uncovered a novel structure that moves oxygen ions through the cell at substantially lower temperatures than previously thought possible. The finding announced this month in Nature Materials may be key to solving fuel cell reliability issues and lead to reduced operating costs in high-performance stationary fuel cells.
Electricity is produced in fuel cells from the electrochemical reaction between a hydrogen-rich fuel and oxygen that produces electric current and water. Research on small fuel cells for cars has dominated the news, but stationary fuel cells are the Goliaths—operating at up to 70 percent efficiency and providing enough electricity—up to 100 megawatts—to power small cities, hospitals, military installations or airports without relying on the electric power grid. Smaller versions are being considered for auxiliary power units in such applications as refrigeration trucks to reduce engine idling.
They are called "solid oxide" fuel cells (SOFCs) because the heart of the cell is a solid electrolyte that transports oxygen ions extracted from air to meet with hydrogen atoms. This alchemy traditionally requires high temperatures—about 850 degrees Celsius in conventional SOFCs—and therefore long startup times, ranging from 45 minutes to eight hours.
The high temperatures necessitate more expensive materials and higher operating costs, so stationary fuel cell research is focused on lowering operating temperatures as well as shortening startup times. The U.S. Department of Energy's goal is to slash the startup time to two minutes.
Chemists at the University of Liverpool fabricated a new oxygen ion electrolyte material of lanthanum, strontium, gallium and oxygen and sent it to the NIST Center for Neutron Research (NCNR) to investigate with collaborators from NIST, the University of Maryland and University College London. Neutrons provide an atomic-scale view of materials so scientists can "see" what is happening at that level.
The oxygen ions in the new materials become mobile at 600 degrees C, much lower than previously studied materials. Researchers suspected the reason lay in the location of the oxygen ions in the crystal framework of the compound. The neutron probes allowed them to determine the basic crystal structure that held the lanthanum, strontium, gallium and oxygen atoms, however the exact nature of the extra oxygen ions was unclear.
NCNR researchers recommended borrowing a method from radio astronomy called maximum entropy analysis. "When astronomers are not able to visualize a specific part of an image because it constitutes such a small part of the total information collected, they utilize a part of applied mathematics called information theory to reconstruct a sharper image," explains NCNR researcher Mark Green. "The combination of neutron diffraction and maximum entropy analysis not only allowed us to determine the location of additional oxygen ions outside of the basic framework, but revealed a new mechanism for ion conduction."
"It allows us to take a fundamentally different approach in the design of future materials, so that we can harness this new mechanism for oxide ion conduction and produce lower operating fuel cells," says Green. "This type of work is very important to us, which is why as part of the NCNR expansion we are developing a new materials diffractometer that will greatly enhance our capabilities in energy related research."
X. Kuang, M.A. Green, H. Niu, P Zajdel, C. Dickinson, J.B. Claridge, L. Jantsky and M.J. Rosseinsky. Interstitial oxide ion conductivity in the layered tetrahedral network melilite structure. Nature Materials, June 2008

