Summary
Industry needs thermodynamic and kinetic data for the development of new materials for sustainable energy applications; expanded and new databases are needed for experimental and computational methods in materials development. The goal of this project is to provide thermodynamic and diffusion mobility databases as an efficient method of storing the wealth of these data, focusing on needs in the energy generation and storage industries.
Description
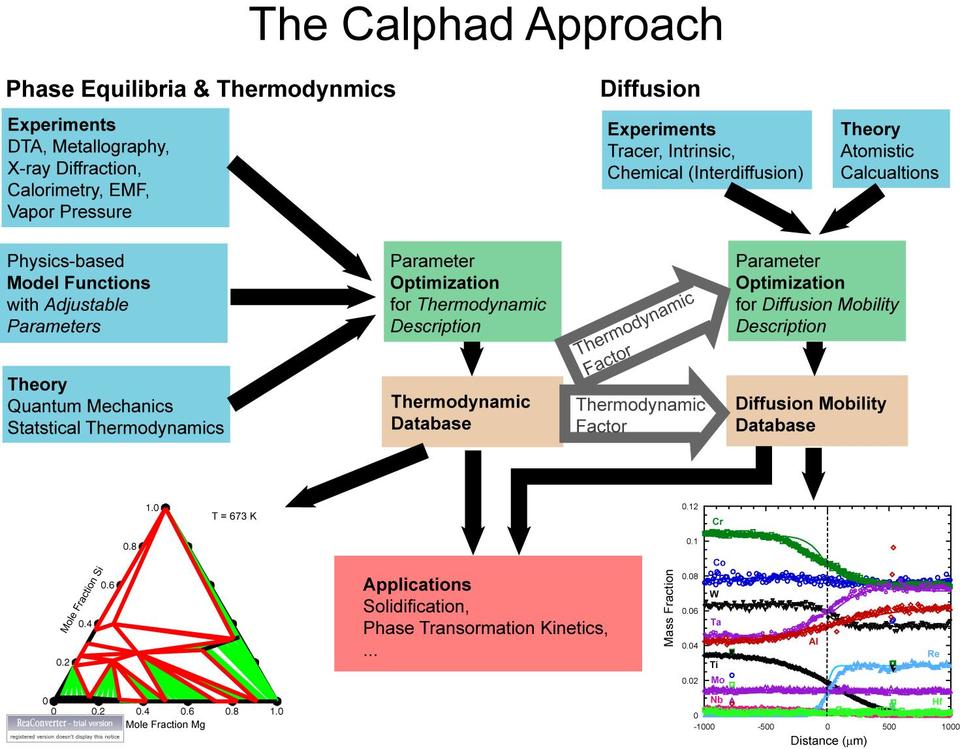
Knowledge of the thermodynamic, phase equilibria and diffusion properties of potential novel materials can greatly accelerate their development. However, the data needed for new, multi-component materials are often not available.
The CALPHAD (Computer Coupling of Phase Diagrams and thermochemistry) approach allows the development of thermodynamic and diffusion mobility databases enabling the extrapolation of higher systems from binary and ternary systems. Phase equilibria calculations using the thermodynamic databases determine the phases present and compositions, as well as enthalpy contents, temperature and concentration dependence of phase boundaries, and enable the coupling of microscopic and macroscopic models.
Going beyond equilibrium properties, these methods can also be applied to the dynamics of materials. Diffusion rates can be expressed as a product of a thermodynamic factor and diffusion mobility. Diffusion mobility functions are optimized with the CALPHAD method using a variety of diffusion data and a given thermodynamic description. These diffusion mobility databases have become crucial in numerical diffusion process simulations for multi-component alloys where composition and temperature-dependent diffusion-coefficient matrices are needed at each point in the material.
Major Accomplishments
Currently NIST is focused on extending the CALPHAD methodology beyond traditional metallic systems as it works to develop improved thermodynamic and diffusion mobility descriptions for photovoltaic and hydrogen storage materials.
Working in conjunction with researchers at the University of Florida, thermodynamic and diffusion mobility descriptions for the Cu-In-Ga-Se system are being developed. This system is critical in the processing of the α-Cu(In,Ga)Se2 (CIGS) photovoltaic absorber material. Advantages of the CIGS-based photovoltaic cells include high efficiency (19.9%), the possibility of direct band gap engineering, high optical absorption coefficient, high radiation resistance, high reliability, and use on flexible substrates. Production costs, however, prevent CIGS from being widely used. To reduce the processing cost requires reducing the current processing times from an average of 30 minutes to less than 3 minutes.
NIST is developing a diffusion mobility database for the Cu-In-Ga-Se system to use with thermodynamic descriptions to enable modeling of a variety of different processing routes. The mobility descriptions are optimized using available measured diffusion coefficients and composition profiles from the literature and growth rate constants measured during in-situ high temperature x-ray diffraction studies by U. Florida and Oak Ridge National Laboratory. Initial comparisons of measured and calculated interdiffusion coefficients for some of the binary intermetallic compounds in the system are shown in the figure below.
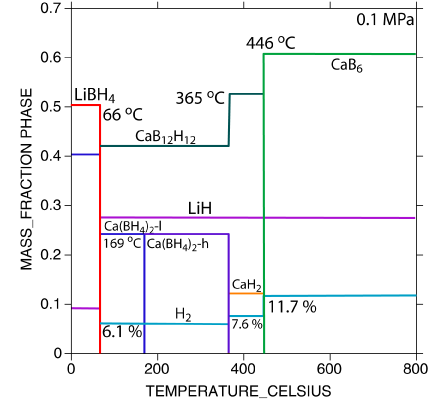
To support materials development efforts in energy storage, NIST has developed thermodynamic databases for Calphad modeling from the available literature for light weight metal hydrides for the Hydrogen Economy, which includes the elements Li, Mg, Ca, B, Si, and H and their respective binary and ternary phases and for Laves phase based hydrogen storage materials.
For the development of the database for light weight metal hydrides experimental data and first-principles calculation results were incorporated into an overall temperature - pressure – composition framework for metal-hydrogen systems with three or more components. Data and functions have been compiled into a consistent database describing the multi-component system. Missing quantities have been identified, and descriptions are being developed in collaboration with MHCoE partners. The Neumann-Kopp rule for the prediction of heat capacities was modified for more accurate predictions for compounds with anion complexes. This database has been used to calculate the reaction paths in multi-component hydrogen systems. These results are the basis for the evaluation of the suitability of promising light weight hydrides and their mixtures that have the potential for a favorable hydrogenation/dehydrogenation temperature and storage capacities suitable for automotive applications. The figure shows the reaction path for a 6LiBH4 + CaH2mixture. Hydrogen is released in multiple steps at different temperatures.
For the Laves phase based hydrogen storage materials databases for four constituent ternary systems (Cr-Ni-Ti, Cr-Ni-Zr, Cr-Ti-Zr and Ni-Ti-Zr) were assembled from descriptions in the literature. Effective ternary compositions were chosen for comparison of the calculated liquid composition paths and their associated solid phase formation sequences with the experimentally observed solidification path of the nine component alloy (Ni, Zr, Ti, V, Co, Mn, Cr, Al, Sn). The features of the ternary systems, Cr-Ni- Zr and Cr-Ti-Zr, are consistent with the observed microstructure, primary solidification of a C14 phase followed by a C15 phase. The intermetallic phases completing solidification are also predicted to occur in some of the four ternaries. The figure shows the liquidus surface of the Cr-Ni-Zr system and the solidification path for the effective composition. The results suggest that development of a full multicomponent Laves phase thermodynamic database will be useful for efficient design of alloys for use as metal hydrides in battery technology.
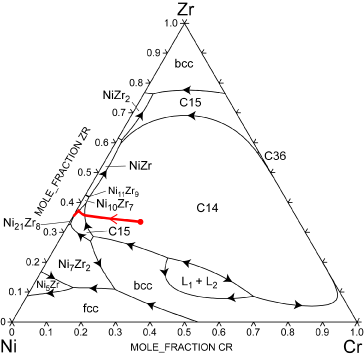
Liquidus surface of the Cr-Ni-Zr system. The solidification path for the effective alloy composition is shown in red.
While extending the CALPHAD methodology to specific materials systems of interest, NIST is also working to establish a set of reference self-diffusion mobilities to enable more efficient construction of multicomponent diffusion mobility databases in the future.Just as the lattice stabilities for the free energy parameters describing the thermodynamics must be self-consistent within a multicompoent system, so must the pure element self-diffusion mobility coefficients. That is, the values defining the self-diffusion of fcc Ni in assessed Ni-Al system should be same as those used in assessed Ni-Cu system.This effort was initiated in the spring of 2009 with a meeting of selected international experts in diffusion mobility modeling, first principle calculation methods as applied to diffusion, and experimental experts. The group is currently preparing a set of recommendations for Ni, Al, Cu, and Fe.
Project Summary (PDF)

