Summary
A variety of nutrients are essential for normal functioning of the human body, and deficiencies can lead to serious illnesses. Accurate measurement of vitamin levels is therefore essential for identifying individuals affected by nutritional deficiencies and for evaluating the potential health benefits associated with dietary supplementation. Previous interlaboratory comparisons have indicated that discrepancies can exist between different assays used to assess nutritional status and that there is a significant need for standardization of these measurements. NIST has developed a series of serum-based SRMs that can be used as control materials by laboratories measuring vitamins (e.g., folate, D, B6) in human serum.
Description
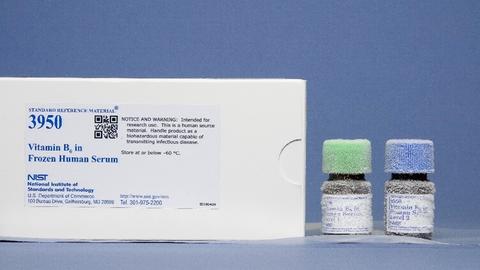
SRM 3950 Vitamin B6 in Frozen Human Serum
Vitamin deficiencies are generally considered rare in the U.S., but a growing body of evidence suggests that certain deficiencies may be more prevalent than originally thought. For example, it has been estimated that 10 % to 15 % of adults over the age of sixty are affected by vitamin B12 deficiency, but most of these individuals lack any overt symptoms. Assessment of nutritional status has been performed by a variety of techniques, including microbiological, enzymatic, immunoassay, and chromatographic approaches. Interlaboratory comparisons have revealed discrepancies in the results obtained by laboratories using different assay methods or even when different laboratories use the same assay for a given analyte. Such variation adds significant complexity to the identification of "normal" ranges for nutritional analytes, and reduces the reliability of vitamin deficiency diagnoses. To improve the accuracy and reliability of nutritional status assessments, NIST is developing a series of serum-based Standard Reference Materials (SRMs).
Major Accomplishments
- SRMs for B6 and vitamin D in serum (SRM 3950, SRM 972a, SRM 2973), as well as for fat-soluble vitamins and carotenoids in serum (SRM 968f), are currently available.
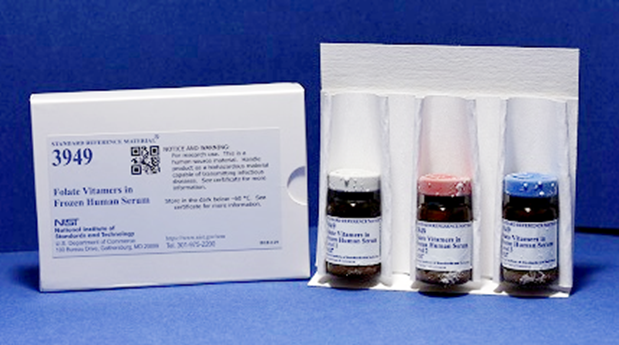
- Candidate material for SRM 3949 has been received and value assignment is in progress.
- Higher-order methods have been developed for these vitamins.
Additional Technical Details
SRM 968f Fat-Soluble Vitamins in Frozen Human Serum was released in 2017 and replaces SRM 968e. SRM 972a Vitamin D Metabolites in Frozen Human Serum is currently available and consists of four different sera. Vitamin D status is generally monitored by measuring levels of 25-hydroxyvitamin D (25OHD). Because vitamin D has two forms, D2 and D3, accurate assessment of vitamin D status should include measurement of both hydroxylated forms (25OHD2 and 25OHD3). The levels of both analytes, as well as those of related compounds 3-epi-25OHD and 24R,25-OH2D3, are characterized in the SRM. SRM 2973 Vitamin D Metabolites in Frozen Human Serum (High Level) contains a higher level of 25OHD3 and is also available.
SRM 3950 Vitamin B6 in Human Serum is another vitamin-related SRM that is currently available and consists of two sera with different analyte concentrations. Vitamin B6 is converted to pyridoxal 5'-phosphate (PLP), the analyte of clinical interest when assessing vitamin B6 dietary status. Although PLP deficiency is believed to be rare, tests for PLP deficiency are not widely performed because of a lack of accepted analytical methodology and because quality control materials are not readily available.
SRM 3949 Folate Vitamers in Frozen Human Serum is currently in development and will consist of three sera with different folate concentrations. Each of these SRMs has been designed in consultation with a panel of experts, and the analyte levels are intended to span the range typically found in patient samples.
Associated Products
- SRM 968f Fat-Soluble Vitamins in Frozen Human Serum
- SRM 972a Vitamin D Metabolites in Frozen Human Serum
- SRM 2973 Vitamin D Metabolites in Frozen Human Serum (High Level)
- SRM 3950 Vitamin B6 in Frozen Human Serum
Associated Publications
1. Camara, J. E., Pritchett, J. S., Daniels, Y. C., Bedner, M., Nelson, M. A., Lowenthal, M. S., Fazili, Z., Pfeiffer, C. M., Phinney, K. W., Sharpless, K. E., Sander, L. C., Lippa, K. A., Yen, J. H., Kuszak, A. J., and Wise, S. A., "Development of an improved standard reference material for folate vitamers in human serum," Analytical and Bioanalytical Chemistry, 415, 809-821 (2023).
2. Boggs, A. S. P., Kilpatrick, L. E., Burdette, C. Q., Tevis, D. S., Fultz, Z. A., Nelson, M. A., Jarrett, J. M., Kemp, J. V., Singh, R. J., Grebe, S. K. G., Wise, S. A., Kassim, B. L., and Long, S. E., "Development of a pregnancy-specific reference material for thyroid biomarkers, vitamin D, and nutritional trace elements in serum," Clinical Chemistry and Laboratory Medicine, 59, 671-679 (2021).
3. Phinney, K. W., Tai, S. S. C., Bedner, M., Camara, J. E., Chia, R. R. C., Sander, L. C., Sharpless, K. E., Wise, S. A., Yen, J. H., Schleicher, R. L., Chaudhary-Webb, M., Maw, K. L., Rahmani, Y., Betz, J. M., Merkel, J., Sempos, C. T., Coates, P. M., Durazo-Arvizu, R. A., Sarafin, K., and Brooks, S. P. J., "Development of an Improved Standard Reference Material for Vitamin D Metabolites in Human Serum," Analytical Chemistry, 89, 4907-4913 (2017).
4. Tai, S. S. C., Nelson, M. A., Bedner, M., Lang, B. E., Phinney, K. W., Sander, L. C., Yen, J. H., Betz, J. M., Sempos, C. T., and Wise, S. A., "Development of Standard Reference Material (SRM) 2973 Vitamin D Metabolites in Frozen Human Serum (High Level)," Journal of Aoac International, 100, 1294-1303 (2017).
5. Bedner, M. and Phinney, K. W., "Development and comparison of three liquid chromatography-atmospheric pressure chemical ionization/mass spectrometry methods for determining vitamin D metabolites in human serum," Journal of Chromatography A, 1240, 132-139 (2012).
6. Phinney, K. W., Bedner, M., Tai, S. S. C., Vamathevan, V. V., Sander, L. C., Sharpless, K. E., Wise, S. A., Yen, J. H., Schleicher, R. L., Chaudhary-Webb, M., Pfeiffer, C. M., Betz, J. M., Coates, P. M., and Picciano, M. F., "Development and Certification of a Standard Reference Material for Vitamin D Metabolites in Human Serum," Analytical Chemistry, 84, 956-962 (2012).
7. Tai, S. S. C., Bedner, M., and Phinney, K. W., "Development of a Candidate Reference Measurement Procedure for the Determination of 25-Hydroxyvitamin D-3 and 25-Hydroxyvitamin D-2 in Human Serum Using Isotope-Dilution Liquid Chromatography-Tandem Mass Spectrometry," Analytical Chemistry, 82, 1942-1948 (2010).
8. Nelson, B. C., Pfeiffer, C. M., Zhang, M., Duewer, D. L., Sharpless, K. E., and Lippa, K. A., "Commutability of NIST SRM 1955 Homocysteine and Folate in Frozen Human Serum with selected total homocysteine immunoassays and enzymatic assays," Clinica Chimica Acta, 395, 99-105 (2008).
9. Thomas, J. B. R., Kline, M. C., Schiller, S. B., Ellerbe, P. M., Sniegoski, L. T., Duewer, D. L., and Sharpless, K. E., "Certification of fat soluble vitamins, carotenoids, and cholesterol in human serum: Standard reference material 968b," Fresenius Journal of Analytical Chemistry, 356, 1-9 (1996).

