Summary
The ability to acquire large amounts of high quality data very rapidly is increasingly important for progress in the biological sciences. Capillary electrophoresis (CE) has a number of advantages that make it well suited to provide the kind of quantitative data that is necessary. However, high throughput CE instruments are complex and expensive, limiting their widespread use. This project is directed at the development of a new approach to multiplexed electrophoresis which is much simpler and less expensive. In the new approach, the bundle of long separation capillaries is replaced by an array of very short electrophoresis channels, and the multi-point optical detection system of the conventional CE approach is replaced by an array of electrical resistors. The new electrophoresis device can be made to fit into the format of a microtiter plate, making it compatible with existing robotic systems for high throughput measurements.
Description
Intended impact:
In the biological sciences, there is an increasing need for measurement tools that can provide high-throughput, high-quality data (consider drug discovery and systems biology, for example). Capillary electrophoresis (CE) is fast, requires only small amounts of sample and reagents, and can be applied to a wide variety of biochemical analysis problems. In addition, capillary array electrophoresis systems have been described for high throughput operation. However, one obstacle to the wider application of high throughput CE is the complexity and high cost of capillary array instruments and techniques.
Objective:
Develop a new method for multiplexed capillary electrophoresis that is simpler and less expensive than conventional technologies.
Goals:
- Determine the simplest possible device that can be used for electrophoretic assays
- Determine parameters and design rules for optimization of device performance
- Develop methods for device fabrication and operation
- Demonstrate use of device for high-throughput biochemical assays
Major Accomplishments
- A method for "detectorless" electrophoresis has been developed.
- A theoretical framework for device and parameter optimization has been developed.
- A 16 channel device has been fabricated and tested.
- The device and method have been demonstrated for high-throughput measurements of kinase activity and inhibition.
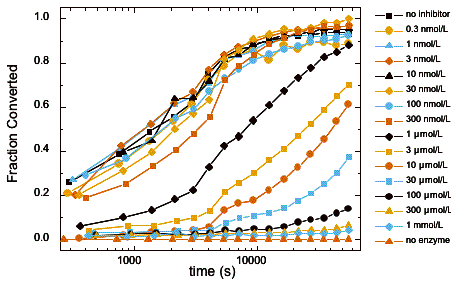
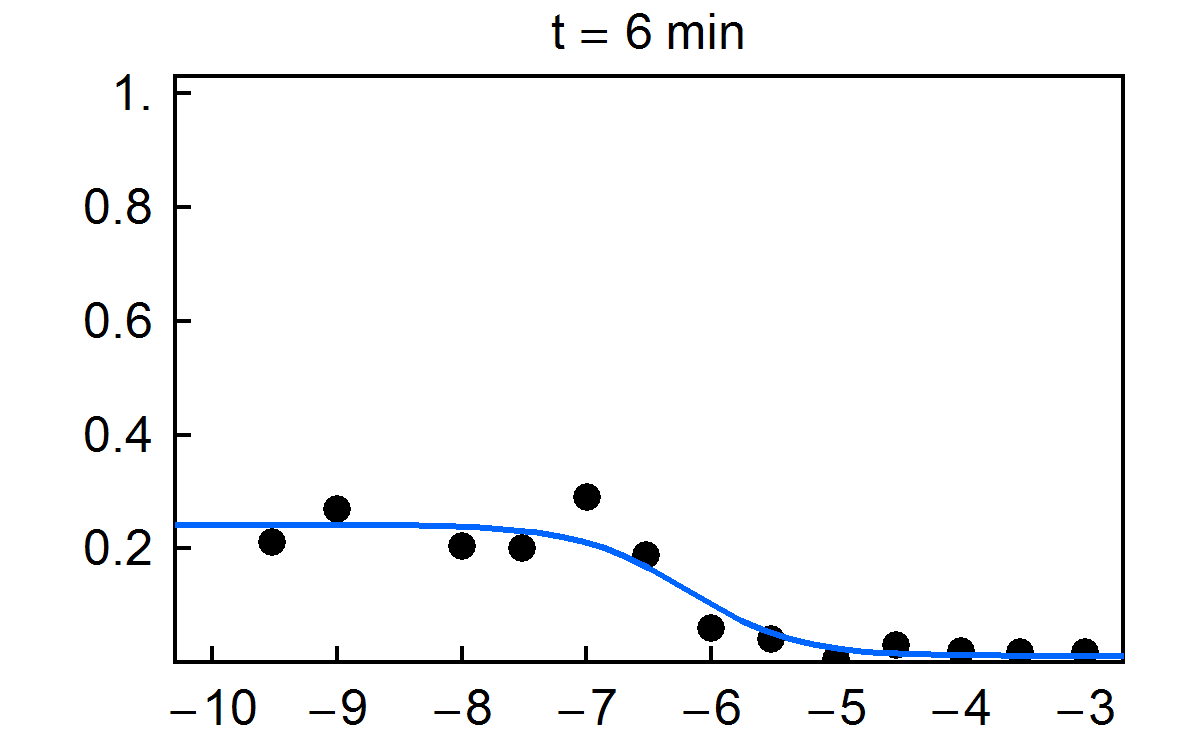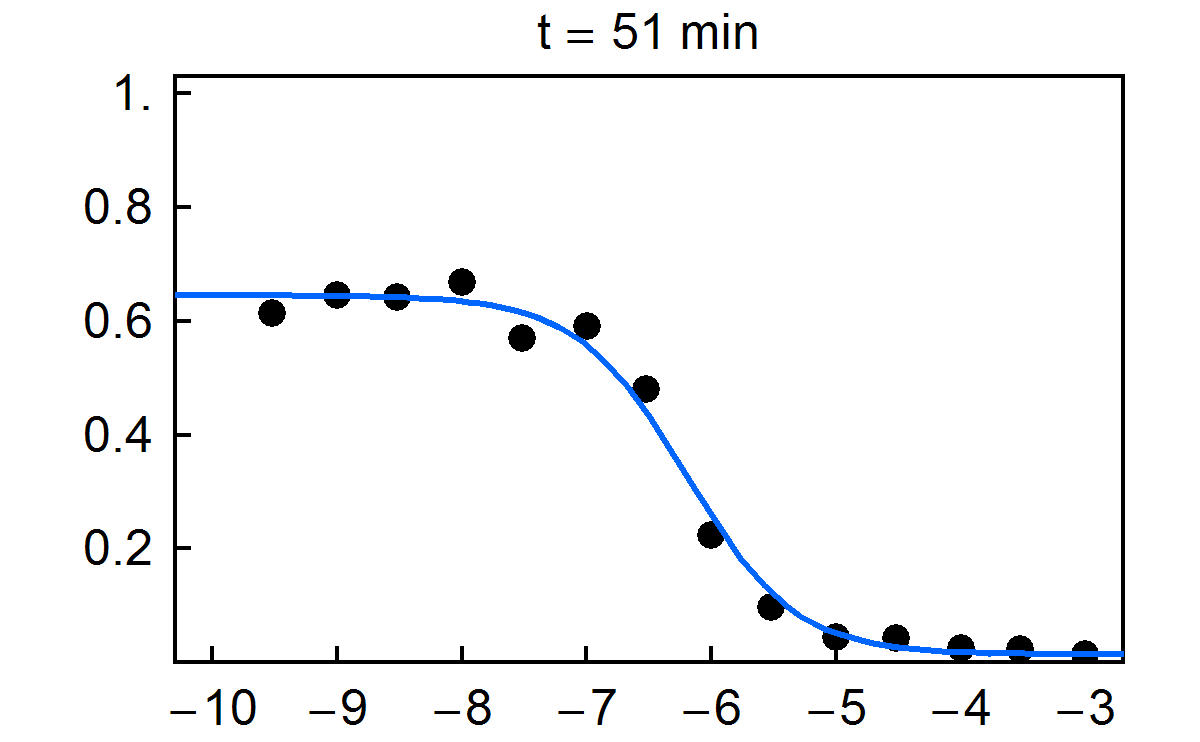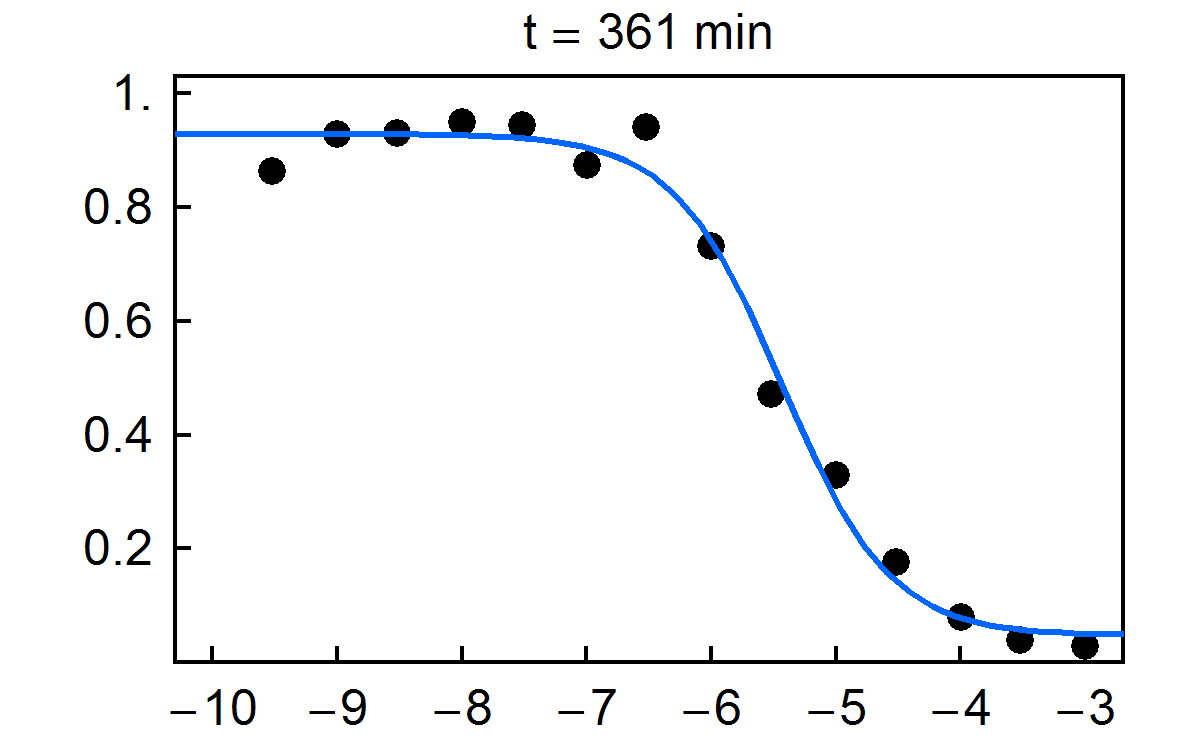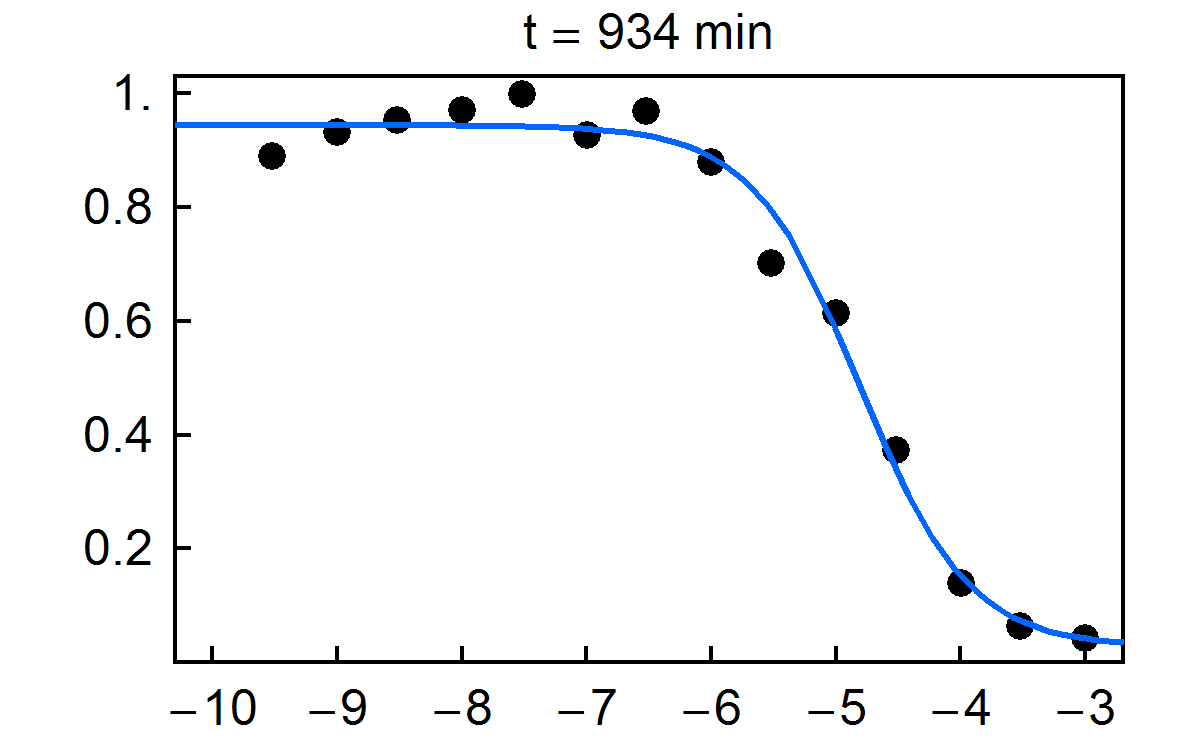
Fraction of substrate converted to product vs. time for multiplexed enzyme inhibition measurement. Final concentrations of the inhibitor H-89 dihydrochloride are given in the figure.
NIST has developed a new approach for multiplexed electrophoretic analysis. This "detectorless" approach is an implementation of the gradient elution moving boundary electrophoresis (GEMBE) technique, previously developed at NIST. With GEMBE, a combination of an electric field and controlled, variable buffer counterflow is used to achieve high resolution separations of multiple analyte species. Typically, the polarity of the applied voltage is set so that the charged analytes are driven by electrophoresis from the sample toward the separation channel. At the beginning of a separation, the counterflow is high so that none of the analytes of interest will enter the channel. Over the course of the separation, the counterflow is gradually reduced so that each analyte, in turn, will enter the channel as a moving boundary or step. For "detectorless" analysis, very short separation channels (< 3 mm) are used, so that only one step at a time is present in each channel. Consequently, measurement of the current through each electrophoresis channel can be used in place of a detector signal, with essentially no detector hardware. The time derivative of the current is then equivalent to a conventional electropherogram. Because of the simplicity of the hardware used, the "detectorless" electrophoresis approach is simple and inexpensive to multiplex; making it ideally suited to high throughput measurements.

