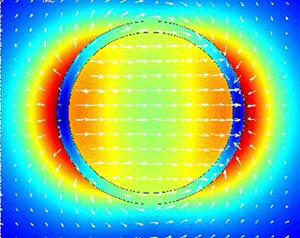
Field within and around a 60 nm radius gold nanoring. Enhanced fields inside the nanoring would make the nanoring a useful container for precision nanoexperiments.
Gold rings suddenly have new cachet, and it has nothing to do with the price of gold or trends in marriage. Moreover, in this case, smaller rings are better.
NIST scientists and collaborators have demonstrated that when a gold ring has a radius of about 60 nanometers, it acquires special properties that could make it a useful container for nanoscale experiments that would provide unusually clear results. Such experiments would be of interest to the drug industry and biochemical researchers, among others.
Metal nanoparticles are attracting research interest because they can absorb and emit certain types of light very efficiently, depending on particle size and shape. In a recent paper, NIST physicists and collaborators in Sweden and Spain documented that gold nanorings have unique optical and electromagnetic properties that can be "tuned" by varying the ratio of the ring radius and wall thickness.
When light is directed at the ring, it excites the electrons at the metal and creates a wave that oscillates in certain ways depending on the wavelength of light and ring geometry. The effect is similar to that of wind hitting a swimming pool, where the resulting wave movement depends on wind velocity and pool shape. The NIST team determined how to synchronize the energy of the incoming light and the pool of electrons, so that they "resonate" on the same wavelength.
This resonance creates a strong and uniform electromagnetic field that oscillates inside the ring cavity. The figure depicts the field within and around a ring with a radius of 60 nm and a wall thickness of 10 nm. The colors indicate the field strength, ranging from blue (weak) to green and yellow (strong and uniform) to red (very strong). The arrows indicate the direction of the field created by the pool of excited electrons.
The field inside a ring with these dimensions is optimized for the near-infrared part of the electromagnetic spectrum. The ring cavity could therefore be a useful container for experiments in which molecules are tested with light, because it would amplify infrared signals. For example, researchers study the chemical bonds in biomolecules like proteins by probing them with lasers and detecting how much of the light is absorbed and re-emitted at specific frequencies. By conducting such experiments inside a nanoring, researchers could obtain amplified infrared signals and clearer results.

