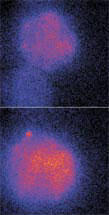
Top: Fluorescent images show two liposomes, one filled with dye and another filled with calcium ions. Bottom: After fusing, the contents of the liposomes mix and react producing an increase in the total fluorescence.
Using laser light as tweezers and a scalpel, scientists from the National Institute of Standards and Technology (NIST) have demonstrated the use of artificial cells as nanovials for ultrasmall volume chemistry. The approach may be useful for faster, cheaper identification of new pharmaceuticals and for studying cellular-level processes. The researchers will report their results in the Sept. 30 edition of Langmuir.
The artificial cells, called liposomes, are tiny spherical containers that self-assemble from natural fats (phospholipids and cholesterol). Measuring micrometers in diameter, the fluid-filled membranes are currently used in cosmetics and for drug delivery.
The NIST team developed an improved method for using liposomes as tiny test tubes for mixing chemicals with volumes measured in trillionths of liters. Their experimental setup allows simultaneous trapping of two liposomes without deforming or stressing their membranes, a problem with some other techniques. They used pairs of infrared lasers ("optical tweezers") to bring two liposomes into contact and a single ultra-violet laser pulse (the "optical scalpel") to fuse the two cells together. Once fused, the contents of the two cells mix and react. One liposome in each pair contained fluorescent dye, and the other contained calcium ions. After the cells merged, fluorescence increased as a result of the reaction between the dye and the ions.
The optical scalpel achieves cleaner fusion and less leakage of contents than the typical technique using pulsed electric fields. The liposomes fully enclose their reactant chemicals, minimizing evaporation. Consequently, the technique also may be useful for quantitative studies of chemical reactions involving samples in the quadrillionths of liters.

