Summary
Nanocalorimetry provides a capability to measure the thermal properties of very small samples and at very fast rates. Materials and interfaces of interest include thin films, nanoparticles, nanowires, advanced gate stacks for microelectronic devices, and biomaterials. The project develops methods, instruments and microfabricated sensor chips and has collaborators working in many areas of materials science and biomaterials.
Description
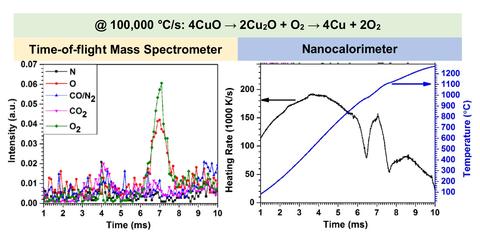
Results from simultaneous nanocalorimeter time-of-flight-mass-spectrometer measurements of the decomposition of copper oxide, showing the thermal curves and the gas species evolved during the experiment. The heating rate was over 100,000 °C/s. The experiment took 10 milliseconds and reached over 1000 °C.
Accurate thermodynamic measurements are essential to understand fundamental properties of materials, providing direct and quantifiable insight into the thermodynamics of thin film reactions and phase transitions. Going forward, new classes of materials may only be synthesized as thin films, a scale at which traditional calorimetric techniques are not useful. The heat capacity, transition temperatures, and specific heat of very small samples (nanogram) can be quantified, including thin films, multilayered films, nanoparticles, polymer coatings, biomaterials, and nanocrystalline and amorphous materials.
A variety of devices have recently been developed for nanocalorimetry, ranging from MEMS based differential scanning calorimeters (DSC) to scanning probe calorimeters. The ongoing development of these new devices requires advances in thermometry, and detailed understanding of the thermal behavior of the materials from which they are made. Discrete sensors for use at modest temperatures are commercially available; extending nanocalorimetry to higher temperatures is important for inclusion of more classes of materials.
Our goal is to establish nanocalorimetry as a quantitative technique by developing MEMS-based nanocalorimeter sensors and instrumentation, calibration procedures, and reference materials for these devices. We perform extensive thermal modeling of our nanocalorimeters to quantify the accuracy and precision of data generated by these devices. The applicability of nanocalorimetry is demonstrated with measurements on industrially relevant materials, such as multilayer thin film structures necessary for next generation integrated circuit devices.
Advanced electronic and optoelectronic materials are used in highly integrated structures such as multilayer thin film stacks. The performance of devices containing such structures is critically dependent on the stability of the thin film interfaces. Nanocalorimetry can determine the stability of multilayer thin film structures by quantifying the thermodynamics of interfacial reactions.
Other applications for NIST nanocalorimeters are the assessment of relaxation in glassy sugars, evaluation of bulk metallic glass stability, and the quantification of reactions (such as nickel-silicide formation) in next generation silicon integrated circuit devices.
Related Publications
|
Feng Yi, Greg Gillen, J Lawrence, Tom P Forbes, Mathew Staymates, David A LaVan “Nanocalorimetry of explosives prepared by inkjet printing” Thermochimica Acta 685, 178510 (2020) Feng Yi and David A. LaVan “Nanocalorimetry: Exploring Materials Faster and Smaller” Applied Physics Reviews 6 031302 (2019) Feng Yi, Lawrence H. Friedman, Richard Chen, David A. LaVan “Sample Pattern and Temperature Distribution in Nanocalorimetry Measurements” Journal of Thermal Analysis and Calorimetry (2019) Feng Yi, Michael D. Grapes, David A. LaVan “Practical Guide to the Design, Fabrication and Calibration of NIST Nanocalorimeters” NIST Journal of Research (2019) Shane Q. Arlington, Feng Yi, David A. LaVan, Timothy P. Weihs “A Nanocalorimetric Study of the Effect of Composition Gradients on Crystallization in Amorphous Cu-Zr Thin Films” AIP Advances 9, 035324 (2019) Emanuel Franke, David A. LaVan, Cynthia A. Volkert “Quantifying DC differential scanning nanocalorimetry for determining heat capacities” Thermochimica Acta 668 116-125 (2018) Michael Grapes, Melissa K Santala, Geoffrey H Campbell, David A LaVan, Timothy P Weihs “A detailed study of the Al3Ni formation reaction using nanocalorimetry” Thermochimica Acta 658 72-83 (2017) Feng Yi, Ana Stevanovic, William A. Osborn, A. Kolmakov and David A. LaVan “Multi-environment Nanocalorimeter with Electrical Contacts for Use in the Scanning Electron Microscope” Materials Horizons 4 1128-1134 (2017) Feng Yi, Jeffery B. DeLisio, Nam Nguyen, Michael R. Zachariah and David A. LaVan “High Heating Rate Decomposition Dynamics of Copper Oxide by Nanocalorimetry-Coupled Time-of-Flight Mass Spectrometry” Chem. Phys. Lett. 689 26-29 (2017) Jeffery B. DeLisio, Feng Yi, David A. LaVan, Michael R. Zachariah “High Heating Rate Reaction Dynamics of Al/CuO Nanolaminates by Nanocalorimetry-Coupled Time-of-Flight Mass Spectrometry” Journal of Physical Chemistry C 121 2771-2777 (2017) Feng Yi, Jeffery B. DeLisio, Michael R. Zachariah, David A. LaVan “Nanocalorimetry coupled Time-of-Flight Mass Spectrometry: Identifying evolved species during high rate thermal measurements” Analytical Chemistry 87 9740-9744 (2015) Feng Yi, Osborn, W.; Betz, J.; LaVan, D.A. “Interactions of Adhesion Materials and Annealing Environment on Resistance and Stability of MEMS Platinum Heaters and Temperature Sensors” JMEMS 24 1185 - 1192 (2015) Michael D. Grapes, Thomas LaGrange, Karsten Woll, Bryan W. Reed, Geoffrey H. Campbell, David A. LaVan and Timothy P. Weihs “In situ transmission electron microscopy investigation of the interfacial reaction between Ni and Al during rapid heating in a nanocalorimeter” APL Materials 2 116102 (2014) Feng Yi and David A. LaVan “Hydrated / Dehydrated Lipid Phase Transitions Measured Using Nanocalorimetry” J. Pharm. Sciences 103 3442-3447 (2014) Michael D. Grapes, Thomas LaGrange, Lawrence Friedman, Bryan W. Reed, Geoffrey H. Campbell, Timothy P. Weihs, David A. LaVan “Combining nanocalorimetry and dynamic transmission electron microscopy for in situ characterization of materials processes under rapid heating and cooling” Rev. Sci. Inst. 85 (2014) Feng Yi and David A. LaVan “Electrospray-assisted Nanocalorimetry Measurements” Thermochimica Acta 569 1-7 (2013) P. Swaminathan, M. Grapes, S.C. Barron, D.A. LaVan and T.P. Weihs, “Studying exothermic reactions in the Ni-Al system at rapid heating rates using a nanocalorimeter” Journal of Applied Physics 113 143509 (2013) Brian G. Burke and David A. LaVan “Laser Heating and Detection of Bilayer Microcantilevers for Non-Contact Thermodynamic Measurements” Applied Physics Letters 102 021916 (2013) B. G. Burke, R. L. Gates, W. A. Osborn and D. A. LaVan "Nanoscale specific heat capacity measurements using optoelectronic bilayer microcantilevers" Applied Physics Letters 101 243112 (2012). Brian Burke and David A. LaVan “Comparison between optical and opto-mechanical measurements with a 10 MHz quadrant avalanche photodiode for detection of microcantilever displacements” Applied Physics B – Lasers and Optics (2012) P. Swaminathan, D.A. LaVan and T.P. Weihs “Dynamics of solidification in Al thin films measured using a nanocalorimeter” J. Appl. Phys. 110: 113519-113524 (2011) Feng Yi and David A. LaVan, “Nanoscale Thermal Analysis for Nanomedicine by Nanocalorimetry” Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology (2011) P. Swaminathan, B.G. Burke, A.E. Holness, B. Wilthan, L. Hanssen, T.P. Weihs, D.A. LaVan, “Optical calibration for Nanocalorimeter Measurements” Thermochimica Acta 522:60-65 (2011) Ravi K. Kummamuru, Lito De La Rama, Liang Hu, Mark D. Vaudin, Mikhail Y. Efremov, Martin L. Green, David A. LaVan, and Leslie H. Allen “Measurement of heat capacity and enthalpy of formation of Nickel Silicide using Nano-calorimetry” Applied Physics Letters 95: 181911 (2009) |
Major Accomplishments
Accomplishments of this project include:
- Developed designs and processes to make nanocalorimeter sensors in the NIST CNST cleanroom
- Studied effect of adhesion metal on heater performance and optimized sensor design
- Studied effect of heater shape and voltage probe spacing and optimized sensor design
- Studied effect of annealing environment on heater performance and optimized the anneal cycle.
- Developed optical calibration tool for rapid sensor calibration
- Developed sample deposition methods for broad classes of materials ranging from nanoparticles to reactive multilayer thin films
- Extended temperature range of measurements with the nanocalorimeter sensors to 1200 °C
- Performed measurements on reactive materials, energetic materials, nanomaterials, thin films, lipids
- Integrated nanocalorimeters into other instruments for simultaneous measurements in the dynamic transmission electron microscope, the scanning electron microscope, the atmospheric pressure XPS at the Advanced Light Source and the time-of-flight mass spectrometer
- Trained many students, post-docs and researchers on nanocalorimetry methods.
- Multiple publications reporting on our advances in the method and detailed measurements of novel materials (see publications below).

