Summary
Our goal is to develop reliable tools and standards for measuring the properties of cells, biomaterials, scaffolds and tissue-engineered constructs. As regenerative medicine moves from a research endeavor to a viable industry that benefits patients and U.S. commerce, reliable methods for assessing product consistency and quality are required.
Description
Tissue Engineering Measurands: Tissue engineering measurands are confusing to discuss and a charting method to improve communication is demonstrated.
Measurement Assurance Strategies for Regenerative Medicine: Reliable methods for assessing product consistency and quality.
Regenerative Medicine: Clinical translation of advanced therapies requires better characterization of products and more robust, reliable, and cost-effective manufacturing processes.
ASTM Committee F04 on Tissue-Engineered Medical Products: ASTM serves a neutral ground where stakeholders can openly discuss and develop standards for measurements of product attributes.
Absorbance Imaging & AI for Assessing Quality of Tissue Engineered Retinal Pigment Epithelium: Measurement of RPE pigmentation as a non-invasive method to assess tissue quality during biomanufacturing.
Combinatorial Cassettes: Combi-cassettes enable 19 tissue-engineered constructs to be placed into one mouse, improving the quantitative rigor of animal tests.
Measuring Cell Viability in Scaffolds: An ASTM working group has been formed to develop a standard test method for assessing viability of cells encapsulated in hydrogel scaffolds.
Scaffold Characterization: Validated, open-source ImageJ plugin (DiameterJ) for automated measurement of nanofiber diameter in scanning electron micrographs.
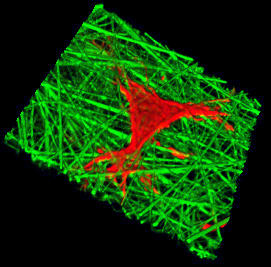
3D Cell-Scaffold Interactions: 3D imaging of cells and scaffolds to provide fundamental principles for design of 3D tissue-engineered constructs.
Scaffold Fabrication: Airbrushed Scaffods & Combinatorial Methods: Methods for fabricating scaffolds and scaffold libraries.
Scaffold Handling Device: A positive and negative air pressure device for efficient and gentle handling of nanofiber scaffolds.
Regenerative medicine represents a new paradigm in human health with the potential to resolve unmet clinical needs through technologies that are specifically developed to modify diseases and enable tissue regeneration or replacement.
There are approximately...
- 30 regenerative medicine products currently on the market
[Regenerative Medicine Annual Industry Report, Alliance for Regenerative Medicine, 2014] - 672 regenerative medicine companies worldwide
[Annual Data Report on Gene & Cellular Therapies & the Regenerative Medicine Sector, Alliance for Regenerative Medicine, 2015] - 631 regenerative medicine clinical trials underway
[Annual Data Report on Gene & Cellular Therapies & the Regenerative Medicine Sector, Alliance for Regenerative Medicine, 2015]
Drivers:
- A survey of pharma by Alliance for Regenerative Medicine (https://alliancerm.org/) found that "product consistency & lack of standards is possibly the single greatest challenge facing the field" for regenerative medicine
[Pharma & Biotech Survey, Alliance for Regenerative Medicine, 2014] - A survey by Regenerative Medicine Foundation (https://regmedfoundation.org/) found that the "lack of reference materials to benchmark measurements and validation criteria for critical assays" is the biggest roadblock for the field
[Hunsberger J, Harrysson O, Shirwaiker R, Starly B, Wysk R, Cohen P, Allickson J, Yoo J, Atala A. Manufacturing road map for tissue engineering and regenerative medicine technologies. Stem Cells Translational Medicine 2015, 4:130-135] - A new Regenerative Medicine roadmap identified "cell attribute measurements" as a primary need to enable reproducible manufacturing of high-quality cells
[Achieving Large-Scale, Cost-Effective, Reproducible Manufacturing of High-Quality Cells: A Technology Roadmap to 2025, National Cell Manufacturing Consortium AMTech, 2016] - The U.S. spends more of its GDP (18%) on healthcare than any other nation, second place is Switzerland at 11% (Organization for Economic Cooperation and Development)
- In 2004, the estimated cost of treatment and lost wages associated with musculoskeletal diseases was $849B (8% of U.S. GDP)
[The Burden of Musculoskeletal Diseases in the United States, American Academy of Orthopeadic Surgeons, 2008] - Diabetes, a major tissue engineering target (pancreatic islets), affects 6 million patients and costs $125B/yr (6% of US healthcare spending)
- In 2004, the estimated cost of treatment and lost wages associated with musculoskeletal diseases was $849B (8% of U.S. GDP)

