Summary
Novel technologies for solar fuel production are expected to increasingly contribute to meeting our nation's environmental and energy needs. Mimicking nature's ability to photochemically split water is attractive because the resulting products, hydrogen and oxygen, are environmentally benign as is the downstream combustion product, water. Recent developments in producing solar fuels include biologically-templated nanoparticle catalysts for hydrogen production and electrodeposited metal-organic compounds for water oxidation. Many details about the assembly chemistry, the identity, and the location of the catalytically active sites are unknown for these nanoscale materials. This project focuses on making fundamental measurements of the reaction chemistry and catalytic intermediates in catalyst nanoarchitectures for water oxidation. It places specific emphasis on the development of constructs that improve the processibility and lifespan of the materials.
Description
A transition to solar fuel will require high-efficiency catalysts that make use of domestic fuel feedstocks with minimal additional energy input. In the past several years, novel water oxidation catalysts from abundant elements have been reported. Catalysts deposited onto a photoactive substrate provide half of a photochemical hydrogen fuel production cycle. Fabricating these composite catalysts into well-defined nanostructures can improve production efficiency and substrate accessibility. The evolution of these systems into the final morphology will be measured using characterization methods with nanoscale spatial resolution.
Soft nanomaterials, including biological macromolecules such as proteins and DNA, are versatile supports for organizing nanostructures and catalysts. Catalysts, including biocatalysts, capable of carrying out the multi-electron chemistry needed to produce solar fuels require multiple chemically distinct catalytic centers. These catalytic centers often involve metal ions in paramagnetic states to maximize reactivity and product distribution. Knowledge of the structure and function of the biosupported catalyst is integral to making further improvements and decreasing biocatalyst development time. Final nanomaterial quality relies on control of the nucleation and growth phases of the material, an aspect that is particularly important in creating metallated biomacromolecules, typically via reduction of metal ions bound to designed biomolecular sites. These processes will benefit from structural elucidation of the bionanomaterials, including measurement of the distances between metal ions in the engineered sites.
Electron paramagnetic resonance (EPR) spectroscopy is a particularly powerful tool for nanoscale systems lacking long-range crystalline order — the regime in which many bionanomaterials fall. Pulsed EPR spectroscopy provides information about the electronic environment of paramagnetic centers through determination of magnetic parameters including the g value, hyperfine couplings, and nuclear quadrupole interactions. Recent advances in pulsed EPR spectroscopy permit detailed interrogation of the magnetic properties and structure of paramagnetic centers in these "soft" bionanomaterials. Combining pulsed EPR spectroscopy with the ability to apply a voltage and to optically excite the assemblies to initiate water oxidation catalysis will permit in situ probing of the catalyst oxidation state, coordination environment, and reaction chemistry during catalytic cycling. In conjunction with an atomic force microscope with scanning electrochemical and Raman microscopy capabilities to map topography, redox processes, and physicochemical changes, we will systematically examine structure and chemistry at the nanoscale to develop tight structure-function maps to help transition these materials into the solar fuels marketplace.
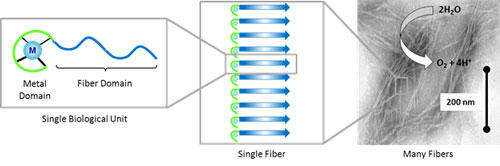
Schematic diagram of how metal-containing protein subunits self-assemble to form fibers that carry out artificial photosynthesis reactions, including water oxidation. Use of biologically-templated nanomaterials is limited by a lack of metrologies to determine the atomic and molecular-level catalyst environment under working conditions, the electronic structure of surface sites on nanoclusters, and the chemical bonding of metal centers in catalytically active assemblies. Measurements to reveal chemical information about the catalyst binding sites and their activity in or on the scaffold are required to reveal the versatility, limitations and applications of these biomimetic assemblies.
Selected Publications
- Affinity of Cu+ for the copper-binding domain of the amyloid-β peptide of alzheimer's disease, H. A. Feaga, R. C. Maduka, M. N. Foster, and V. A. Szalai, Inorganic Chemistry 50, 1614-1618 (2011).
- Cu K-edge X-ray absorption spectroscopy reveals differential copper coordination within amyloid-β oligomers compared to amyloid-β monomers, J. Shearer, P. E. Callan, T. Tran, and V. A. Szalai, Chem. Commun. 46, 9137-9139 (2010).
- Fluorescence of unmodified oligonucleotides: A tool to probe G-quadruplex DNA structure, M. A. Mendez and V. A. Szalai, Biopolymers 91, 841-850 (2009).

