Summary
We develop quantitative methods for the analysis of macromolecules having complex architectures and nanoparticles having organic functionalities. Our approaches leverage new concepts in mass spectrometry and numerical optimization, and help industry discover, develop, and understand the complex materials that form the basis of emerging technologies.
Description
Bringing new products to market requires measurements for rapid, quantitative assessment of composition and structure. To this end, NIST has developed a series of mass spectrometry-based tools that decrease the time necessary to achieve measurement success while producing more accurate results. The development of our tools and techniques has been guided by the needs of U.S. industry through our series of annual workshops as well as our active participation in the Versailles Project on Advanced Materials and Standards (VAMAS).
| Examples of our unique tool set include the MassSpectator computer code for automated, unbiased peak integration of mass spectral data; software for numerical optimization of instrument parameters; sample preparation methods employing aerosol deposition of analytes to ensure robust, reproducible experimental results; and a mass spectrometer that enables melting of samples during analysis. Our Taylor's Expansion Method for determining absolute molecular mass distribution of macromolecules and nanoparticles is a milestone in quantitative mass spectrometry of polydisperse materials. The unique mass spectrometer, custom-made sample preparation devices, and computer code developed in our laboratories are themselves prototypes for potential future products. |
 |
Major Accomplishments
|
 |
Macromolecules
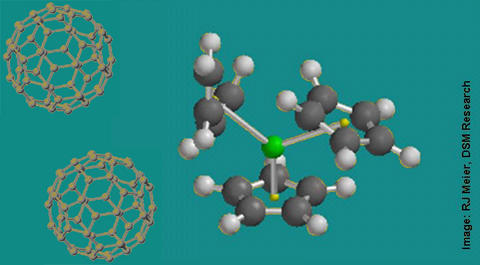
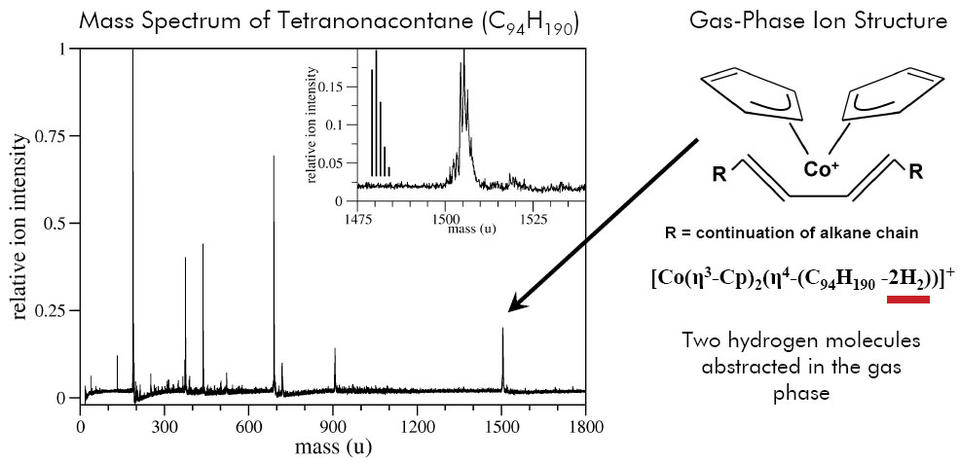
The inability to produce intact, high mass alkane and polyolefin ions in the gas phase has been a long standing barrier to the mass spectrometric analysis of saturated hydrocarbons, thereby limiting its broader use in the petroleum and the polyolefin industries. Analyte fragments are only able to tell part of the story of a material's composition and structure. Density functional calculations have shown that bare metal cationization, the typical way to Reactive MALDI of a high mass alkane ionize unsaturated hydrocarbon materials,is impossible without chain scission when applied to saturated hydrocarbons under typical desorption conditions.
Recently the use of organometallic cations has gained prominence as a way to create intact gas phase alkanes and polyolefins. In our recent work, organometallic cationization of high mass alkanes and polyolefins is applied for the first time to matrix-assisted laser desorption ionization (MALDI). This has several important benefits.
The difficulty with the direct application of organometallic ionization to MALDI is that the most commonly used precursor is highly volatile and rapidly boils off into the vacuum of the mass spectrometer. To overcome this limitation, NIST created a novel MALDI matrix that intercalates an organometallic compound into the fullerene (C60) face centered cubic lattice. The entrainment of the organometallic in the matrix prevents it from boiling into the vacuum. UV laser ablation converts the precursor organometallic into the organometallic cation necessary to charge the polyolefin. In turn, this organometallic cation locally unsaturates the polyolefin in a controlled fashion leading to an intact alkane as shown in the figure above. We call this method reactive MALDI.
The results presented here open up new avenues of reactive MALDI for analytes that are traditionally hard to produce as intact, gas phase ions. It is further envisioned that reactive MALDI may be used to selectively ionize only certain chemical species in a complex mixture, or certain chemical moieties on heterogeneous macromolecules. The joining of organometallic gas phase chemistry with the needs of the macromolecular mass spectrometry community offers a host of undiscovered possibilities of potential benefit to U.S. industry. For example, selective ionization of components of polymer mixtures could decrease the complexity of the resultant mass spectrum.
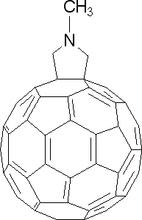
NanoparticlesRecently, work has begun on determining the number of functional groups grafted onto fullerenes. For example, the number of methyl pyridine groups that can be added to the fullerene cage (shown below) ranges from one to nine, with significant differences in number coming from slight changes in preparation chemistry. Mass spectrometry allows for the quantitative determination of the number of ligands appended to the fullerene cage. This can have a major effect on hybrid materials properties such as the number of re-enforcing fullerenes that can be put into an epoxy matrix. This measurement has proven to be an indispensable tool to aid the synthetic chemist. |
 |

