How Do You Know the Age of Fossils and Other Old Things?

The universe offers a few kinds of natural clocks. We measure the frequency of light absorbed by atoms to keep very accurate time and precisely define the second. On the other end of the spectrum, nature gives us a variety of “nuclear” clocks. We can use them to count not seconds but instead thousands, millions and even billions of years. We call this method of timekeeping radiometric dating. While it’s usually not accurate down to the second, it can give us a good idea of the ages of things such as rocks, fossils and remains of plants and animals.
Radiometric dating is based on the phenomenon of radioactivity. Atoms of radioactive elements, such as uranium, have so much energy that their cores, or nuclei, are literally flying apart, shedding motes of their matter in a process called decay. Through our observations and understanding of physics, we know that each radioactive element decays at its own steady rate. As they shed particles, the atoms change into atoms of other elements, their “progeny,” until they finally become stable. Uranium, for instance, goes through a long decay chain that eventually ends with lead. Through observations and understanding of chemistry, scientists know that lead-and-uranium-containing minerals form with certain proportions of each element. As the uranium decays, the ratios of the elements will change. By combining our knowledge of chemistry and physics, we can use the rate at which uranium atoms decay and observations of the ratios of the elements today to figure out how old the minerals are.
We often talk about radioactive decay in terms of half-lives. A half-life is how much time it takes half of the radioactive atoms in a sample to decay. The half-lives of radioactive atoms that we have calculated cover durations from vanishingly short, 23 yoctoseconds (trillionths of a trillionth of a second) for helium-7, to far longer than the present age of the universe, as estimated for atomic hydrogen.
Atoms of an element often appear in several different forms, or isotopes, which have slightly different weights or masses. The half-life of uranium-238, uranium’s most abundant and longest-lived isotope, is approximately 4.47 billion years. Using uranium-238 and uranium-235, which has a half-life of 704 million years, scientists were able to determine the age of minerals found on Earth, the Moon and inside meteorites to help us estimate that Earth formed 4.54 billion years ago, give or take 50 million years.
Geologists use the radiometric dating of uranium and the isotopes of other radioactive elements, combined with observations of fossils and sediment layers, to chart the course of Earth’s natural history and the evolution of life.
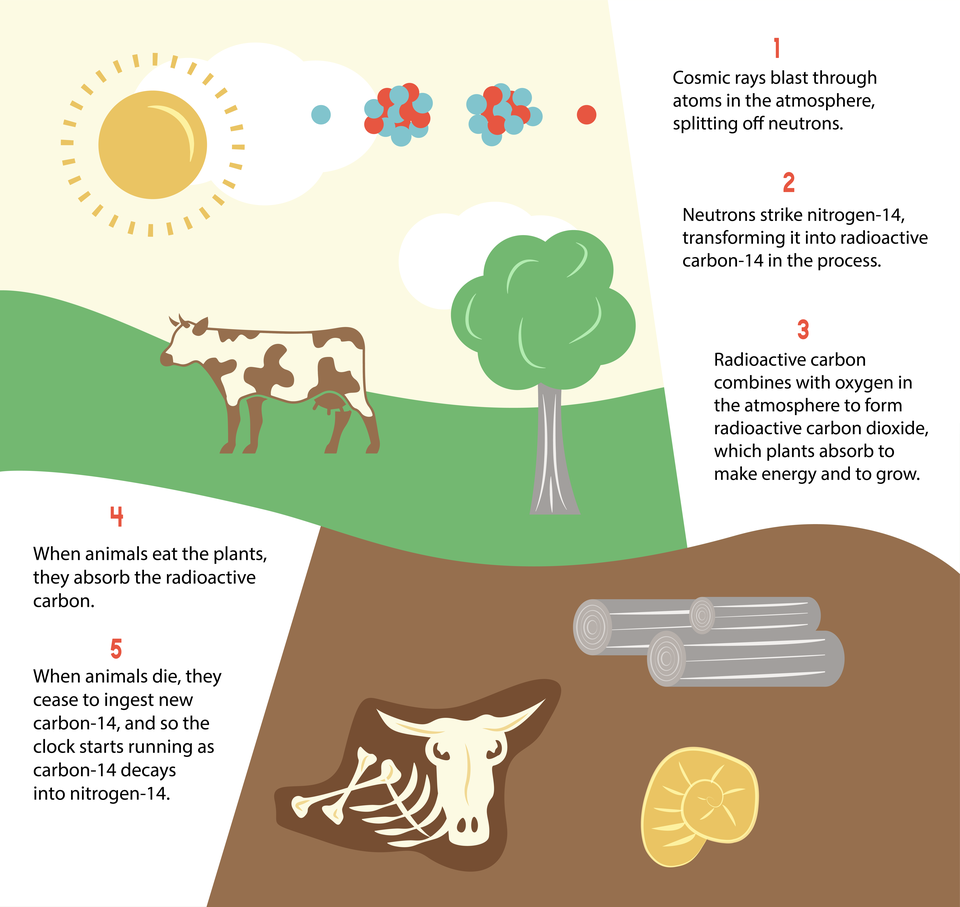
To measure the age of plant and animal remains from the more recent past, scientists use a radioactive isotope of carbon, called carbon-14, as their clock. As carbon-14 decays, with a half-life of about 5,730 years, it becomes nitrogen-14. Using this clock, they have dated bones, campfires and other objects as old as 60,000 years, and in some cases even older.
The way the carbon-14 gets into the tissues of plants and animals starts with cosmic rays. Cosmic rays blast through atoms in the atmosphere, splitting off neutrons. These neutrons combine with nitrogen-14 atoms in the atmosphere to create radioactive carbon-14. This radioactive carbon combines with oxygen in the atmosphere to form radioactive carbon dioxide, which plants absorb to make energy and to grow. When animals eat the plants, they absorb the radioactive carbon. When plants and animals die, they cease to take in new carbon-14 and so start the clock running as the carbon-14 decays into nitrogen-14.
Scientists have verified the accuracy of carbon-14 dating by studying the rings of trees and using historical objects, like samples from the tombs of Egyptian pharaohs whose date of reign we know.
By revealing the ages of things, radiometric dating has made it a lot easier to read the history written in the rocks and artifacts buried in the Earth, giving us a sense of the vast time that it took for our planet to take shape and for life and eventually us to evolve. We can only wonder at what it will reveal about the past next.

