Certification of Standard Reference Material 2393 for the CAG repeats of Huntington Disease
Summary
Huntington disease is a neurodegenerative disease of midlife onset that produces choreic movements and cognitive decline, often accompanied by psychiatric changes that affects approximately 1 in 10,000 individuals. Inheritance is autosomal dominant with clinical manifestations associated with expansion of a polymorphic trinucleotide (CAG) repeat.Description
This Standard Reference Material (SRM) is intended primarily for use in the value assignment of the number of Huntington's Disease (CAG) trinucleotide repeats contained in a human genomic material. The six component genomic DNA materials are from DNA extracted from cell lines derived from Huntington's Disease samples (Coriell Cell Repositories, Coriell Institute for Medical Research, Camden , NJ).
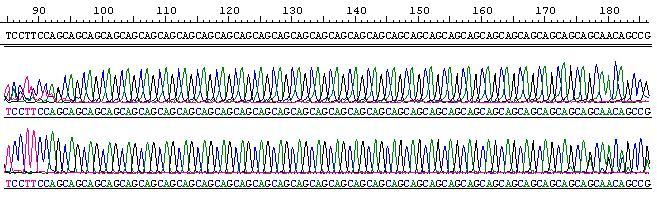
The number of (CAG) repeats was determined by counting the number of (CAG) repeat units after Sanger sequencing. These values were confirmed by fragment analysis (genotyping).
The components include the following alleles: 15, 17, 29, 35, 36, 39, 40, 45, 50, & 75.
These selected alleles represent the "normal" to "Juvenile onset" stages of the disease.
For more information, please see the Certificate for SRM 2393 or NIST Tech Beat article on SRM 2393
Major Accomplishments
- Certification of SRM 2393 - CAG Repeat Length in Huntington's Disease
- Established fluorescent genotyping methods suitable for capillary electrophoresis analysis.
- Established an enhanced method for the sequencing of the Huntington Alleles:
-
- Selection of novel sequencing PCR primers
- Enhancement of the Sanger sequencing reaction mix
- Selection of cell lines having the appropriate alleles to span the Huntington disease state from normal to juvenile onset:
-
- Isolation and sequencing of the individual alleles
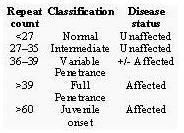
Example of one normal allele and one intermediate allele
Forward and Reverse Sequencing of 29 allele